
This final section of my paraphrase of Hoermann et al’s landmark 2016 article articulates the shift from the limited, older TSH-T4 paradigm to the new paradigm that includes T3 hormone.
Interrelational Measures and Emerging New Concepts of Thyroid Homeostasis
[Paraphrase begins]
TSH can play a useful role in screening patients who do not have any hypothyroid or hyperthyroid symptoms. However, it cannot at the same time assume the second role of being a reliable diagnostic tool (a gold standard) for defining true “euthyroid” status within the TSH range.
We should therefore scale back the common practice of clinical interpretation of TSH as a single biomarker in isolation from the thyroid hormones.
As discussed above, the TSH reference intervals are not defined with statistical certainty even in a healthy population. From a homeostatic perspective, all three parameters of TSH, FT4, and FT3 must be viewed together.
We would like to propose that an individual patient’s thyroid hormone levels should be measured in a state of thyroid health before the onset of disease or thyroidectomy. This would provide better understanding of the dynamic adaptation in hormone levels in relation to each other across these state changes.
One could even create an individual’s own “archival” thyroid hormone levels to serve as a reference point in case thyroid disease arises in the future. The set point may help guide dosage of thyroid medications.
Where full archival data is unavailable for a patient, one may attempt to reconstruct the individual’s set point from multiple TSH-T4 pairs over time, yet using the TSH-T4 relationship alone has two critical disadvantages: it fails to deliver targets for FT3 levels, and it is not sensitive to variations in deiodinase activity involved in converting T4 into T3.
However, it is important to remember that the archival set point in health cannot be used as an equivalent target in the state of disease.
The hormone equilibrium within a single person will shift considerably from health to disease, and it will shift once again under the artificial conditions of hormone replacement therapy. These important changes in variables require flexible adjustments to hormone levels that take into consideration the influence of external hormone medications.
We cannot assume a stable “log-linear” TSH-T4 relationship across the entire functional range of thyroid hormone parameters.
The standard log-linear relationship assumes that TSH will always rise by X units as T4 drops by Y units. The linear relationship is roughly accurate, most of the time, in the “open loop” situation of a hypothyroid patient under LT4 therapy. (The feedback loop is no longer a closed loop system because external thyroid hormones are adding to the system.)
However, the log-linear relationship between TSH and T4 becomes progressively damped toward the TSH range considered “euthyroid.” (As one can see in the graph below, the TSH-FT4 line flattens almost to a horizontal level as TSH comes closer to the top of its reference range.)

Within the euthyroid range, TSH is no longer the dominant factor that expresses control. Other factors begin to dominate over TSH control, such as Free T4 and T3 levels and ratios. The thyroid hormone system attempts to achieve relational stability by fine-tuning responses among all three hormones based on the needs of the situation.
Cross-sectional studies of populations show this pattern quite convincingly, but it is difficult to interpret longitudinal data (over time) because most patients studied are under LT4 treatment. Medical treatment by LT4 has an artificial dose-related influence on the relationship between hormones. Therefore it cannot demonstrate the natural mutual influence among hormones over time.
In particular, especially among patients without a thyroid gland, response to LT4 therapy can be extremely variable and unpredictable from patient to patient taking the same dose, making it unreasonable to apply to all patients a pre-set healthy reference range for all hormone levels.
In healthy people with a HPT axis that is not interfered with by thyroid hormone dosing, Free T3 levels are not directly related to TSH levels. This lack of TSH-T3 relationship in health indicates that their T3 level is stabilized independently from TSH control.
However, in patients on LT4-therapy, FT3 is not only unstable and much more variable, but is very closely related to TSH level.
In thyroid therapy, the FT3 falls as TSH rises in reference, which is not seen in the healthy HPT axis outside of thyroid therapy. Thyroidless patients often fail to achieve mean FT3 levels found in healthy controls within the euthyroid TSH range, achieving them slightly below the TSH reference, instead. [See Figure 1.]

This major difference between healthy people and LT4-treated patients challenges the standard treatment goal of applying inflexible TSH-FT4 setpoints to patients on therapy. Instead, the FT3 and TSH levels should be interpreted dynamically in relation to each other because they have an unnatural relationship in therapy.
Thyroid treatment should not rule out the goal of stabilizing or optimizing Free T3 levels, since this was the target of the HPT axis prior to therapy.
Absolute Free T3 levels are extremely important on their own, and the Free T3 level has an important role to play in the TSH-T4 relationship as well.
Thyroid hormone equilibrium, especially FT3, can easily fall outside of optimal reference ranges during LT4 therapy.
When patients ingest LT4 hormone without producing an appropriate level of T3 hormone to accompany the T4 load on the system, the thyroid hormone equilibrium will attempt to rebalance itself, but will sometimes fail to do so, ending up in non-optimal and abnormal configurations. This is quite different from problems arising from malabsorption of LT4 medication due to GI health or drug interference.
In some LT4 treated patients, low Free T3 concentrations will persist in spite of suppressed TSH levels and higher than normal Free T4 levels. A significant minority of thyroidless patients on LT4 therapy are unable to raise their Free T3 levels into reference because excess LT4 ingestion and absorption impairs the conversion of T4 into T3.
(In Gullo’s 2011 study, the percentage of athyreotic patients with FT3 serum levels lower than the normal range was 8.6% in males and 16.4% in females, and 15.2% lower overall. Among all treated patients, 29.6% had a significantly lower Free T3:T4 ratio. See figure below from Gullo et al, 2011.)
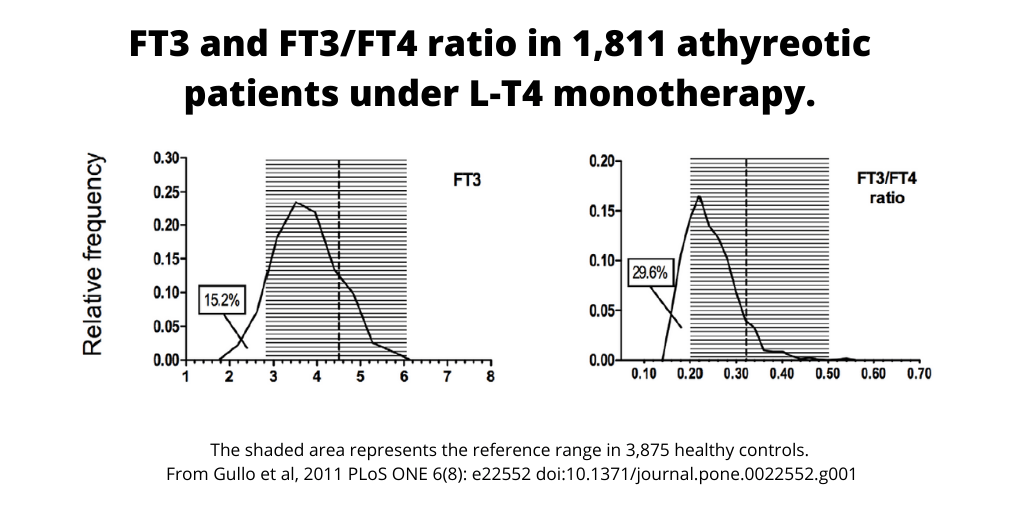
This biochemical imbalance is clinically relevant, affecting tissue T3 levels and health outcomes. Research has shown that targeting only the euthyroid TSH range during therapy was unable to raise resting energy expenditure (REE) in women treated with LT4 compared to healthy people, and only rising FT3 levels were related with rising REE. Another recent study proved that hypothyroid patients on the standard LT4 therapy cannot expect to fully recover from hypothyroidism to the level found in the healthy population.
An important treatment goal is to achieve homeostatic equilibrium based on individually-euthyroid FT3, FT4 and TSH levels and ratios, not just population-wide euthyroid TSH levels.
Conclusion
TSH has a biological role as a controlling element within a biological system where it can partner with healthy thyroid tissue. However, when the homeostatic equilibrium is disturbed by different environmental stimuli, one can’t depend on a univariate (one-variable) statistical concept of a TSH-based euthyroid range to determine everyone within that range as “normal” and euthyroid.
Currently, the TSH plays three roles in medical practice:
- as a sensitive screening test,
- as an accurate diagnostic tool, and
- as a target for therapy.
However, these roles must be separated.
TSH secretion does not play all three roles in normal physiology. Significant biological variation exists between individuals as well as within individuals over time. We see TSH, FT3 and FT4 shift their equilibria and set points even within the population with completely healthy thyroid glands. Homeostatic interrelationships change mainly to protect FT3 stability, and therefore we must also recognize FT3 as the target of thyroid homeostasis. TSH should be interpreted in relationship to Free T3 and Free T4 levels.
An aid to therapy adjustment could be the use of an individual’s archival TSH, FT4 and FT3 levels during a formerly euthyroid state, keeping in mind that the HPT axis will shift in therapy in response to thyroid hormone dosing, as described above.
[End of paraphrase]
All parts of the paraphrase
- Part 1 General introduction
- Part 2 Normality in TSH and thyroid hormones
- Part 3 Shifting relationships between hormones
- Part 4 The new thyroid paradigm
REFERENCES
Andersen, S., Pedersen, K. M., Bruun, N. H., & Laurberg, P. (2002). Narrow Individual Variations in Serum T4 and T3 in Normal Subjects: A Clue to the Understanding of Subclinical Thyroid Disease. The Journal of Clinical Endocrinology & Metabolism, 87(3), 1068–1072. https://doi.org/10.1210/jcem.87.3.8165
Gullo, D., Latina, A., Frasca, F., Le Moli, R., Pellegriti, G., & Vigneri, R. (2011). Levothyroxine Monotherapy Cannot Guarantee Euthyroidism in All Athyreotic Patients. PLoS ONE, 6(8). https://doi.org/10.1371/journal.pone.0022552
Hoermann, R., Midgley, J. E. M., Giacobino, A., Eckl, W. A., Wahl, H. G., Dietrich, J. W., & Larisch, R. (2014). Homeostatic equilibria between free thyroid hormones and pituitary thyrotropin are modulated by various influences including age, body mass index and treatment. Clinical Endocrinology, 81(6), 907–915. https://doi.org/10.1111/cen.12527
Hoermann, R., Midgley, J. E. M., Larisch, R., & Dietrich, J. W. (2015). Homeostatic Control of the Thyroid–Pituitary Axis: Perspectives for Diagnosis and Treatment. Frontiers in Endocrinology, 6. https://doi.org/10.3389/fendo.2015.00177
Hoermann, R., Midgley, J. E. M., Larisch, R., & Dietrich, J. W. (2016a). Relational Stability in the Expression of Normality, Variation, and Control of Thyroid Function. Frontiers in Endocrinology, 7. https://doi.org/10.3389/fendo.2016.00142
Hoermann, R., Midgley, J. E. M., Larisch, R., & Dietrich, J. W. (2016b). Relational Stability of Thyroid Hormones in Euthyroid Subjects and Patients with Autoimmune Thyroid Disease. European Thyroid Journal, 5(3), 171–179. https://doi.org/10.1159/000447967
Russell, W., Harrison, R. F., Smith, N., Darzy, K., Shalet, S., Weetman, A. P., & Ross, R. J. (2008). Free Triiodothyronine Has a Distinct Circadian Rhythm That Is Delayed but Parallels Thyrotropin Levels. The Journal of Clinical Endocrinology & Metabolism, 93(6), 2300–2306. https://doi.org/10.1210/jc.2007-2674

Leave a public reply here, on our website.