
At the end of January 2020, Alberta Precision Laboratories established a new policy of cancelling patient-pay Reverse T3 (RT3) tests on standard laboratory test orders.
One memo in circulation explains the justifications based on RT3 silence in the 2012 and 2014 thyroid clinical guidelines. The memo was issued by Dr. Hossein Sadrzadeh, Section Chief, Clinical Biochemistry, South Sector. It was reviewed by himself and Leland Baskin.
Another memo instructs laboratory staff on procedures, reviewed and approved by Dr. Hossein Sadrzadeh and two other doctors.
In this post, I will first briefly explain what the Reverse T3 hormone is, since it is often misunderstood by patients and doctors.
I’ll then reveal the amazing new scientific breakthroughs that reveal the activity of Reverse T3 on receptors on the cell membrane. As of 2019, science now indicates that RT3 has significant health implications for many diseases, including cancer and cardiovascular disease, so controlling RT3 offers potential health cost savings.
Then I’ll discuss the unethical tradition RT3 research, namely, the routine exclusion of treated thyroid patients from most of the studies of critical illnesses in which RT3 is elevated.
I’ll explain how the test has been useful in clinical practice to adjust thyroid therapy, to aid recovery from illness, and potentially prevent illness by preventing unnecessary NTIS, often characterized by RT3 elevation and FT3 loss despite a normal or low TSH.
Finally, I’ll outline the processes that have been involved in RT3 testing in Alberta. We can make them more efficient and cost-effective for all.
Forward-thinking Laboratory chiefs really should think twice about cancelling this patient-pay Reverse T3 test. Allow the test to continue improving human health in our province.
Reverse T3 and Nonthyroidal Illness
Reverse T3 (RT3) is a thyroid hormone naturally made in our bodies every day from the metabolism of the most abundant thyroid hormone, thyroxine (T4).
When a person has an acute or chronic illness, either in the hospital or in outpatient care, Reverse T3 levels elevate abnormally in relation to the prohormone T4 from which it is produced, diverting metabolism away from the production of T3, the most vital thyroid hormone active in the cells of every organ and tissue of the human body.
This well-known condition in which RT3 elevates is called “nonthyroidal illness syndrome” (NTIS). It has also been called “euthyroid sick syndrome” because the TSH paradoxically does not elevate as thyroid hormones drop. Its most accurate name, however, is “Low T3 syndrome,” because the Low T3 is the most common and worrisome feature.
During the process of NTIS, the RT3 levels may reduce to high-normal range as T4 drops lower in a chronic illness. Yet even a normal-range RT3 will remain abnormally elevated given a lower T4 concentration.
Graphs and models of the phases of nonthyroidal illness differ because different health conditions present with slightly different processes.

The RT3 test is a confirmation of the nature of the process, a feature distinguishing it from other conditions which may result in low T3, such as underdose or poor T4-T3 conversion due to genetics. It is the low T3 concentrations that most often remain independently prognostic of death and morbidity (Rhee et al, 2015). The thyroid hormone alterations seen in NTIS can powerfully influence the progress of all diseases, from surgeries and critical injuries to chronic diseases of the cardiovascular system, kidney, and liver, as well as cancers, and it occurs at any age (Langouche et al, 2019).
Current scientific consensus is that the “acute phase,” the initial drop in T3 and rise in RT3, is likely beneficial and protective in the context of a critical illness. Here, the RT3 elevation is a confirmation that a person is undergoing NTIS. As the body lowers metabolic rate and energy expenditure, the TSH does not elevate to signal a crisis of thyroid hormone loss.

At a crucial turning point in the crisis of illness, when T4 levels are low enough, the body detects that it is time to suddenly raise TSH and encourage the healthy thyroid gland to pump out more T4, and especially a higher percentage of T3. An increase in thyroid hormone blood levels may be delayed because the higher rate of hormone inactivation continues for a while, but the secretion prevents a further fall. In this way, “A rise in TSH levels precedes the onset of recovery from severe illness.” (Van den Berghe et al, 2014).
Sometimes, the TSH elevates significantly above reference (Baloch et al, 2003). This is a biochemical pattern that temporarily mimics overt or subclinical hypothyroidism, but the TSH stimulation is effective when the gland is fully functional. This assertive “push” from the TSH to stimulate the thyroid restores the T3 and T4 hormone deficit, and the patient recovers health as they recover thyroid hormones.

The danger to health is not necessarily in the acute phase when T3 drops. Instead, it is at the turning point between “acute” and “chronic” NTIS.
The greatest danger is:
1) Not having a healthy enough thyroid gland to stimulate, due to preexisting primary hypothyroidism, and
2) Not having enough of a rise in TSH at the right time, due to preexisting central hypothyroidism.
3) Not having any guidelines in place to assess the presence of NTIS outside the hospital using its unique indicators such as RT3 in the context of TSH, FT4, and FT3 and clinical presentation.
4) Not conducting research on risk or prevention of death in patients with known conditions 1) and/or 2).
Not all NTIS illnesses present with an acute phase first. Some gradually encroach and become chronic. Therefore, prevention of chronic NTIS is aided by having health care mechanisms in place to assist in the identification of NTIS even outside of the hospital setting so that the illness can be dealt with before it endangers life (RT3 testing can assist).
RT3 is assessed in context.
Reverse T3 is a troubleshooting test.
Critics of the RT3 test as of 2018 readily admitted that RT3 is useful to distinguish cases of nonthyroidal illness (NTIS), also called “euthyroid sick syndrome” (ESS) from those whose metabolism is unaffected by NTIS:
- “We believe [RT3] can potentially be used to help the differential diagnosis between hypothyroidism and euthyroid sick syndrome. Reverse T3 should always be analyzed in combination with TSH, T4, and free T3 with consideration of the patient’s clinical context.” (Gomez-Lima & Burman, 2018)
This is one of the very few wise statements that Gomez-Lima and Burman have made in their criticism. Our best thyroid doctors in Alberta who order the test and interpret it scientifically could have said the same thing.
Kalra and Khandelwal call on doctors to discover ways of identifying genuine “tissue hypothyroidism” within therapy (2011) so that patients’ complaints of unresolved hypothyroidism can be resolved. If anything can create tissue hypothyroidism, it’s inactivation of T3 and T4 that occurs at the tissue level in NTIS, essentially throwing our medication in the metabolic dustbin.
Reverse T3 testing, in context, also provides a way of distinguishing permanent genetic flaws in thyroid metabolism from temporary, yet life-risking nonthyroidal illnesses.
RT3 is an active hormone.
One of the common reasons given for the dismissal of Reverse T3 testing is that it was believed to be an “inactive” hormone.
As of 2019, this belief has been proven false.
Reverse T3 is now known to be an active thyroid hormone in the same way that T4 is an active hormone.
Reverse T3 and T4 are both inactive at the thyroid hormone receptors in the nucleus of the cell where T3 reigns, but RT3 and T4 are powerfully active at a receptor on the cell surface, the “integrin αVβ3” receptor. (Hercbergs, 2019)

Its activity at this receptor in Cancer, cardiovascular diseases, stroke, and infections is now being revealed.
1. Cancer
In their article “Action of Reverse T3 on Cancer Cells,” Lin and colleagues wrote,
“RT3 is shown in the present study to stimulate proliferation of several types of cancer cells. Thus, rT3 – regarded as a genomically inactive thyroid hormone analog – can, like T4, modulate cell division in tumor cells. The nanomolar concentration of rT3 tested here can be achieved clinically.”
RT3 activity can be pathological in the context of cancer, but it can be cheaply and painlessly reduced by replacing T4 with T3 hormone in persons who are at risk (Hercbergs, 2019). It’s a compassionate approach that requires neither surgery nor chemicals foreign to the human body, and it extends life expectancy by years, not just months.
2. Cardiovascular diseases and microvascular chest pain
As for vascular effects of RT3, in 2019 Davis et al explained that its activity, added to T4’s activity, at this receptor, is involved in angiogenesis — the creation of new tiny blood vessels — and can influence blood vessel health.
This RT3-activated cell membrane receptor stimulates “vascular growth factor,” vascular gene transcription, and “endothelial cell migration” (the endothelium is a layer within our blood vessels), among other things. As it activates similar mechanisms, RT3 may be involved in “endothelial dysfunction,” a process implicated in microvascular angina — a type of chest pain that cannot be detected by the usual cardiovascular tests in an Emergency department (Cannon et al, 2009; see a list in Table 1 in Davis et al, 2018).
3. Blood clotting
According to Davis et al, 2019, this RT3-activated receptor is also found on platelets (small discs of cytoplasm involved in blood clotting). Controlling RT3 levels may therefore be involved in managing blood loss in emergencies and preventing strokes, as RT3 and T4 cause human platelets to clump together and create clots. In contrast, T3 hormone does not have this effect on platelets.
4. Infections
Finally, RT3 is also implicated in the management of infections. The integrin avB3 receptor is involved in “phagocytosis” of bacteria. Phagocytosis is a major mechanism used to remove pathogens. (Davis et al, 2019)
So, why are we cancelling this patient-pay Reverse T3 test?
How many costly emergency visits with mysterious chest pain could be prevented as they are diagnosed in part by testing RT3 levels and other hormones?
How many unnecessary tests for mysterious health conditions would be prevented by discovering that RT3 is not inappropriately elevated given the patient’s FT4 levels?
How might adjustments in treated thyroid patients’ T4, T3, and RT3 levels assist in the mitigation of chronic diseases like cancer, cardiovascular diseases, and infections?
Risk: Thyroid patients’ continued exclusion

Thyroid therapy guidelines do not discuss Reverse T3 testing.
Their silence is taken to mean that the test is irrelevant to thyroid disease management. This is an incorrect assumption.
Silence exists in this field for the wrong reason — a long tradition of unethically excluding a vulnerable population from RT3 and NTIS research.
We don’t know about RT3 dynamics in treated hypothyroid patients because researchers have excluded this population from almost all of their NTIS experiments and retrospective analyses.
Why? Because it’s called “nonthyroidal illness” and we already have a “thyroidal” illness. It’s more interesting to study the paradox of NTIS in patients with healthy thyroids. It is also potentially disruptive of the status quo to study thyroid patients’ NTIS comprehensively.
A very small experiment with six male thyroid patients on LT4 (Synthroid) was done in 2004 (Wadwekar et al). It showed that RT3 rises and both T4 and T3 drop, and the characteristic NTIS pattern occurs over 7 days. But was it a steeper rise and a gentler drop than in thyroid-healthy people going through the same surgeries? There were no healthy controls in the study to compare the degree of difference. Therefore, we don’t know.
These six men had thyroid remnants, given the way that T3 and T4 responded so quickly and vigorously to TSH. They all recovered. Their average FT3: average FT4 ratios were robust before their surgeries, evidence that these were “good converters” of their LT4 thyroid dosage, not vulnerable “poor converters” (Midgley et al, 2015).

Wadwekar’s tiny study on six male thyroid patients tells us nothing about health risk for various types of thyroid patients on various types of therapy. The study was meant to reassure people about the safety of Synthroid and that neglect of any change in dose (or therapy type) could be entirely harmless in six cases. On what basis was “no change” so glibly recommended if no experiments were done to see what happens when the T4 dosage was changed or when some T3 replaced T4 at the turning point? Why are thyroid patients the only people asking these obvious questions?
Risk: Unscientific assumptions
Insufficient experimental basis exists for claims that RT3 testing is irrelevant or useless for the treated thyroid patient population.
There is no reason to assume that LT4 monotherapy is superior to other therapies because it entails lower risk of death in NTIS, and yet thyroid scientists recommend that “Levothyroxine treatment should be continued” without any alteration during NTIS (Fliers et al, 2015).
Critics such as Gomez-Lima and Burman cite Burmeister’s 1995 article extensively to strengthen their dismissal of RT3 testing in thyroid therapy. But the findings of this old study can hardly be applied to the 8 patients who were on LT4 therapy within their study of 246 patients (3% of their population). Burmeister found that “normal RT3” was a common finding in these 8 patients, but admitted his patient population was not large or diverse enough to judge.
Instead of expanding on the tiny number of thyroid patients in these NTIS studies, comparative therapy studies are conducted outside the urgent context of NTIS. There, therapy choice does not immediately influence extreme clinical endpoints such as death or recovery, and few arguments can be made on the basis of health outcomes.
Instead of examining patients already on thyroid therapy in NTIS, studies of T3 therapy in this context are performed on research subjects who have healthy thyroids. Too large or too small doses are used, or too late when diseases have passed the point of no return. Then studies conclude that T3 therapy is not beneficial in NTIS — without verifying if this is true for thyroid disabled people as well.
Scientists also remain willfully ignorant about the prevalence of chronically inflated RT3 levels in a random sampling of LT4 patients who do not have any detectable NTIS (yet). We know that illness causes these biochemical alterations, but how do these alterations cause or worsen illness? What is the average number of months or years that a patient’s Free T3 levels remain chronically low, with or without symptoms, before the onset of elevated RT3 and NTIS?
Indeed, the 2012 guidelines admit that when TSH is normalized on LT4 therapy, “T3 levels may be in the lower reference range and occasionally mildly subnormal” (Garber et al). The acceptance of low T3 in the context of LT4 thyroid therapy is downright shocking. It shows the low priority placed on the thyroid hormone that is most essential for health.
Studies have shown in great detail that a significant percentage of thyroidless treated LT4 patients endures the key biochemical feature of NTIS, a low T3 (Gullo et al, 2011; ). Often such studies omit measures of the RT3 hormone. How can you be sure their hormones are not distorted by illness if you didn’t measure RT3?
If living thyroid gland tissue stimulated by TSH is essential to natural recovery from NTIS (Fliers et al, 2015), what happens to people who have had a total thyroidectomy, or whose gland has been utterly fibrosed by Hashimoto’s, or completely shrivelled by Atrophic thyroiditis? How is a rise in TSH supposed to help them if there is no gland it can stimulate to quickly restore hormone levels, especially the T3 deficit?
And what about people with mild or severe central hypothyroidism, which hinders TSH production? How well do they fare? Will these vulnerable patients be first in line when endocrinologists decide to “investigate treatment with recombinant human TSH” in NTIS? (Fliers et al, 2015)
Our exclusion from research means that the generalizations about RT3 levels and health risk in the untreated, thyroid-healthy population cannot be transferred to patients in the context of thyroid therapy.
Risk: Undetected Chronic NTIS

The cancellation of a patient-pay RT3 test puts at risk the detection and confirmation of many chronic diseases in treated thyroid patients outside the hospital setting.
Only when a condition is severe enough will it manifest in an RT3 level that is inappropriately high, not necessarily above reference, but compared to the FT4. And of course, the low T3 on a chronic basis is a sign of a metabolic failure. It’s not supposed to be chronic.
In some chronic health conditions like heart failure, liver failure, and cancer, the low T3 and inappropriately higher RT3 may occur gradually and undetected. No crisis occurs that sends the patient to hospital in the early stages. The chronic health condition may have a very delayed diagnosis.
The clinical presentation may not fit a doctor’s experience or training, or symptoms may be dismissed as not extreme enough to warrant testing through the health care system. However, the thyroid patient may be able to persuade the doctor to order a test that costs neither the health care system nor should it cost their ego or career.
Let’s consider a similar diagnostic difficulty in thyroid care. Endocrinologists have complained that central hypothyroidism is very difficult to diagnose. Why? Because it can’t be diagnosed by TSH or T4 in isolation, but must be diagnosed but using these two tests together and interpreting them in light of scientific knowledge. Screening and diagnosis is hindered by overreliance on TSH, by policies that prevent FT4 measurement when TSH is normal (Beck-Peccoz, 2017).
It is likewise the case that the relationship between RT3 and other hormones in NTIS are almost impossible to discern in the chronically symptomatic ambulatory thyroid patient because of restrictive testing policies. While such restrictions make sense for people who have not yet been diagnosed and have no thyroid symptoms, it is not logical if the patient is symptomatic while already on thyroid hormone therapy. These policies block our only escape from illness through a diagnosis that will free us from a maladaptive therapy.
When TSH remains normal in undetected NTIS, doctors may incorrectly assume thyroid hormones are normal under the surface. However, in NTIS, TSH circadian rhythm is almost lost, and the night-time thyroidal secretion of T3 is almost lost. The resulting discordance between TSH and low thyroid hormones results in “a presentation resembling central hypothyroidism” in which TSH secretion is abnormally low given low T3 and T4 thyroid hormones (Van den Berghe et al, 2014).
Consider the case of the treated thyroid patient whose chronic symptoms are being dismissed because TSH is anywhere within range, and both thyroid hormones are within range but free T3 is very low, and the doctor cannot detect the presence of a nonthyroidal illness. Their TSH may be abnormally lowered by NTIS, and a self-pay RT3 test could confirm its presence. There may be no other way to pry the grasping fingers of this doctor off the TSH Idol. TSH is untrustworthy in central hypothyroidism, and NTIS induces Central hypo.
Risk: Chronic NTIS with higher T4 levels
A study in 2018 discovered that those who fare the worst in chronic NTIS have high T4 levels concurrently with low T3. (Ataoğlu et al, 2018)
In light of studies like these that investigate T3:T4 ratios in disease outcomes, we should ask this question:
How are LT4-dosed patients faring in chronic disease, when their T4 is usually significantly higher and their T3 usually significantly lower than the healthy population mean when TSH is normalized? (Midgley et al, 2015)
Benefit: Even RT3 “normal” results are still useful
RT3 does not need to be elevated to remain a useful diagnostic tool. It just needs to be inappropriately elevated in relation to T4 to signal a metabolic imbalance.
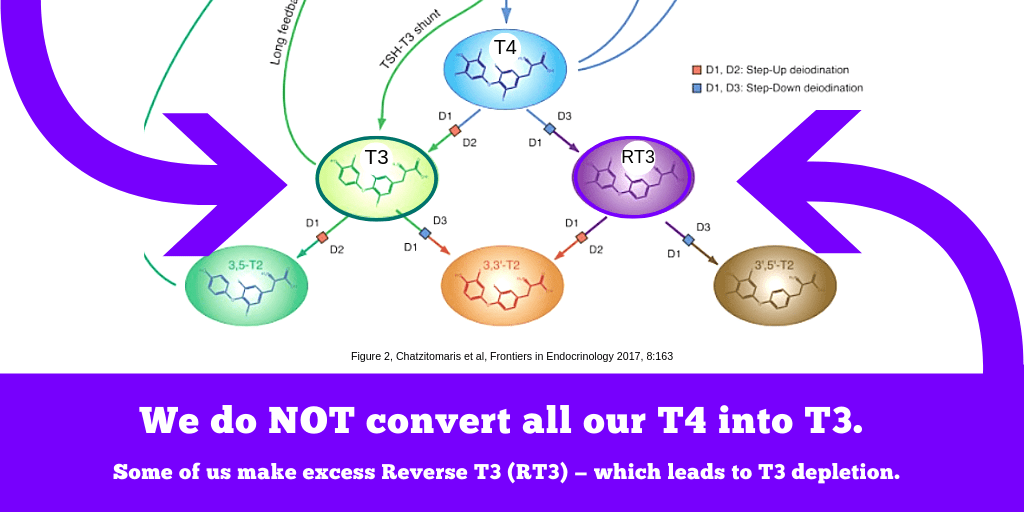
As an illness progresses to a chronic phase and Free T4 levels fall and remain low, Reverse T3 may normalize because there is less T4 from which it can be derived. Nevertheless, even in the chronic phase, when Reverse T3 levels are significantly higher in their range than the Free T4 level, one can discern that a high rate of T4-RT3 conversion is still occurring.
Similarly, the diagnosis of central hypothyroidism is not impossible when TSH is normal or even when T4 is normal. Central hypothyroidism exists with “low, or even low–normal, free T4 with inappropriately low/normal TSH.” (Persani et al, 2019) NTIS, likewise, can exist with inappropriately high or high-normal RT3.
The inappropriateness of RT3 to FT4 can be judged in a similar way as it is for the TSH-FT4 relationship in a Central hypo diagnosis. Just as there are graphs showing the normal TSH-T4 relationship, there is likely a normal reference range for both hormones T4 and RT3. In health, they can be expected to mirror each other’s rise and fall. Allowing for a margin of error expected when comparing two tests using two laboratory test technologies, RT3 levels can still be indicative of illness when they are significantly higher in reference than the FT4 in its reference.
Free T3 aids further in the interpretation of RT3 in the diagnosis of NTIS. Chronically low T3 levels can contribute to the maintenance of higher RT3 concentrations. Deiodinase Type 1’s main role is to clear RT3 from blood. Reduced T3 slows down the rate at which Deiodinase type 1 clears RT3 because this deiodinase’s activity is upregulated by T3 and slowed down by an excess of RT3 to process. (Maia et al, 2011).
Benefit: RT3 testing aids therapy adjustment
Our thyroid patient community (thousands of patients in social media groups) appears to know more about the dynamics of RT3 in therapy than the therapy guideline-writers do.
Our best thyroid doctors will suggest that the patient pay for Reverse T3 when they see patterns in Free T3 and Free T4 that are puzzling in light of our doses and clinical presentation.
We see in our group’s clinical experience that if our Free T4 levels are above our individual metabolic set point even within reference range, the suboptimal FT3 continues and the high RT3 continues. And the illness lingers.
Having a normal TSH does not ensure our health or safety because the TSH is a local tissue response, no longer an essential or central regulator. Dosing is in charge, and Deiodinases are in charge. But the glands that regulate TSH cannot sense the excess T3 in our kidneys or the deficiency in our left elbow.
In contrast, Reverse T3 is a net response from all tissues that exchange thyroid hormones with blood. It can signal when we have too much thyroid hormone in blood for all our tissues, on average, beyond the pituitary and hypothalamus.
Wise thyroid doctors and patients have learned by experience that if we reduce the T4 dose and replace it with an appropriate amount of T3 hormone, the RT3 levels reduce. Doctors may measure RT3 several weeks later to see the effect of the dose adjustment.
Considerate doctors will ask the patient first, to check whether the patient can handle the financial cost for a mere RT3 verification test. The FT3 level and FT3/FT4 ratio is the more powerful indicator.
In context, the optional second RT3 test provides confirmation that T4 conversion is shifting less to RT3 and more towards T3 again and we are better metabolizing our T4 medication. More importantly, as this shift occurs, the patient usually experiences healing and relief from chronic symptoms as Free T3 rises within reference range, which accords with the evidence in Larisch et al, 2018.
Cost-recovery and laboratory efficiency
Presumably, the patient-pay Reverse T3 test was operated on a cost recovery basis.
Calgary Laboratory Services used to charge between $50 and $60 per Reverse T3 test. The patient would pay for the RT3 test by credit card at the laboratory before the blood draw.
Patients would normally have Reverse T3 tested concurrently with TSH, Free T3 and Free T4, since the test must be interpreted in the context of thyroid hormone concentrations.
The tube of blood for the RT3 test would be frozen and shipped, first to another Canadian laboratory, who would then ship it to either LabCorp or Quest Diagnostics in the US.
Upon arrival at the laboratory, the blood would be tested by the highest quality method in thyroid hormone testing, LC-MS, which exceeds the accuracy of routine Free T4 tests.
After a few weeks, the ordering physician would obtain the result, after having had time to ponder the TSH, Free T3 and Free T4 without the RT3 information.
The test involved the willing collaboration between the patient and the test-ordering doctor. The likelihood was high that this test’s results would be taken into account by both parties, since the paying patient had an interest in holding the physician to account.
Therefore, the burden on the Alberta laboratory system was minimal since they had no equipment costs, just minimal collection costs plus shipping.
If the price did not cover the processing labor, materials, or shipping, the price could be raised to cover the cost, rather than the test cancelled.
Clearly, the shipping could be optimized to go directly to the US laboratory so it is less costly and results are not as delayed.
The Calgary and South laboratory locations already have systems and trained staff in place to follow this procedure.
Patients have been travelling down to Calgary from Edmonton and the north just to have access to this patient-pay test when they were denied access in their own region.
Conclusion
Reconsider this decision. Be an innovator in advancing health.
Do not close off access to this patient-pay RT3 test option.
- It is an active hormone.
- It is a hormone with effects on multiple diseases
- It goes against no prohibition in guidelines to test this
- Its clinical use leads thyroid therapy forward as guidelines fall behind
- It enables physicians to optimize symptomatic patients’ therapy
- Those who order and pay for the test choose it and use it wisely
- We can make it even more cost effective and efficient than it was
Guidelines often fall behind research. Research often falls behind innovative clinical practice. Clinicians have discovered ways of enhancing health in spite of the published literature’s silence.
The cost to human health, and the cost to the advancement of many areas of medicine is too much for us to bear.
Yours sincerely,
Thyroid Patients Canada.
- Tania S. Smith

Leave a public reply here, on our website.