
As I’ve outlined in a previous post, central hypothyroidism (CeH) is a failure or compromise in the hypothalamus and/or pituitary gland that causes defective TSH or inappropriately low TSH secretion.
There, I echoed the complaints of world-leading experts in the field, Beck-Peccoz and Persani, who point out why it is so difficult to diagnose central hypothyroidism.
If even thyroid scientists complain that it’s difficult to diagnose central hypothyroidism before thyroid therapy, it may be almost impossible for the medical system to begin to see central hypothyroidism for the first time during thyroid therapy.
In this post, I outline the diagnostic problem, then offer diagnostic solutions from scientific publications.
Fortunately, published graphs and free analytical tools are available to aid in diagnosis of central hypothyroidism in people who have already been diagnosed and are being treated for primary hypothyroidism.
COPYRIGHT NOTICE: Reproduction of a copyrighted article’s data, graphics, and quotations falls within US copyright law as “fair use” and within Canadian copyright law as “fair dealing.” — “A fair use is any copying of copyrighted material done for a limited and “transformative” purpose, such as to comment upon, criticize, or parody a copyrighted work. Such uses can be done without permission from the copyright owner” (Stanford University).
The problem of diagnostic blindness
Our medical system has been blinded by gland-centric and mutually-exclusive definitions of hypothyroidism that make it seem impossible to have more than one type of hypothyroidism at the same time.
Central hypo has been unfairly defined by the exclusion of primary hypothyroidism, and diagnostic guides assume pre-treatment conditions.
It is conventionally diagnosed by low FT4 and a normal or low TSH in the presence of an
“otherwise normal thyroid gland”
(Beck-Peccoz et al, 2017)
.
Another set of scientists define it this way:
“Central hypothyroidism (CeH) is a disorder characterized by defective thyroid hormone production due to insufficient stimulation by thyrotropin (TSH) of an otherwise normal thyroid gland.”
(Persani et al, 2019)
Why must any reasonable person presume the existence of “an otherwise normal thyroid gland”?
Does this mean that in all cases of CeH, the thyroid gland is free of disease or dysfunction? Of course not.
There is no law of nature saying that a thyroid gland and pituitary gland can’t both fail, or that a thyroid can’t fail first, then a hypothalamus or pituitary can fail next.
A skeptic will naturally think “what are the chances of that?” and soon after that, they’ll ask “Where is the research on that?”
Imagine what happens to newborn babies born with pituitary TSH secretion failures (congenital central hypothyroidism) in a country where only TSH, not FT4, is tested in the neonatal heel-prick screening test. Incidence rates will be low.
The failure to screen for a disease makes its incidence rates fall extremely low, but ignorance and neglect is not a cure.
The medical field has made it seem impossible for a person to have both organ failures at the same time, so the chance of finding incidence rates and research on this overlap is bound to be slim.
Once a person is placed on thyroid therapy and the doctor has already categorized them as a “primary hypothyroid” patient whose thyroid gland has failed, how many physicians will even consider Central hypothyroidism (central hypo, CeH) as an additional, overlapping diagnosis later on in the future?
The saying “better the devil you know than the devil you don’t” applies here. Few physicians will blame the devil they don’t know (central hypo), preferring to blame the devil they know (primary hypo).
It’s very tempting for each generation to believe they know everything there is to know about a medical condition. Treatment guidelines can’t yet explain the vast majority of LT4 monotherapy treatment failures, so they have suggested that the physician can blame the patient for “somatization” disorder (Jonklaas et al, 2014) or noncompliance with treatment if the TSH drops for no good reason.
“Once TSH has normalized with treatment, it should be checked annually unless a new indication arises.
This confirms adequacy of treatment dose and compliance with therapy.
(Ministry of health, British Columbia, Canada)
A single hormone can determine whether the patient is compliant with therapy? Is TSH the “eye in the sky,” a patient surveillance system? This is not evidence based medicine, it’s magical thinking that makes the physician feel like they are in control.
For all we know, the phenomenon of “dissatisfied LT4 patients with a normalized TSH” consists of a certain percentage of patients with undiagnosed TSH secretion inhibition, but researchers have not yet been willing to entertain such a hypothesis.
“How dare you accuse the TSH of lying during thyroid therapy!”
It seems like this is what many physicians and scientists would say to a physician or patient who suspects the TSH of misbehavior during thyroid therapy.
The “target” of thyroid therapy is a normalized TSH, according to consensus guidelines. Therefore, it is presumed that TSH can’t lie during therapy.
Can thyroid treatment guidelines prevent the pituitary gland from failing? Of course not, that belief would be ridiculous.
Nevertheless, after the thyroid fails, physicians are expected to put blind faith in the accuracy of TSH hormone concentrations during treatment. They must have no doubt that TSH concentrations represent body-wide thyroid status even though it is known to be a product of pituitary-specific T4 and T3 transport, metabolism and signaling. This attributes to the TSH omniscience about thyroid hormone status in distant parts of the body. If TSH is omniscient, it’s not too much of a stretch to believe in TSH infallibility.
The premise of TSH infallibility is not only illogical, but unethical and harmful. It is like a judge taking sides and defending the TSH when the patient’s body and thyroid hormones accuse the TSH of lying.
During primary hypothyroid therapy,
- The pituitary’s TSH secretion rate becomes a false idol that blinds physicians to its true vulnerability. All other hormone secretions in the human body are permitted to fail, and all other illnesses may be present, as long as physicians imagine that they can’t compromise pituitary TSH secretion. (This makes no sense, because scientists tell us that chronic “nonthyroidal illness syndrome” (NTIS), and some drugs for nonthyroidal illnesses like diabetes and epilepsy, and Graves’ TSH receptor antibodies also found in a percentage of hypothyroid patients, can compromise pituitary TSH secretion.)
- The TSH concentration has been given the veto power to deny the patient’s hypothyroid symptoms and signs of tissue hypothyroidism. This is not evidence-based medicine, this is evidence-biased medicine, in which one data point is believed to be “more accurate” even when it is inconsistent with all other relevant data points and the patient’s experience and testimony.
- When the pituitary TSH is at fault but the case against TSH fails due to lack of knowledge or evidence, the patient is doomed to be punished with an underdose that induces tissue hypothyroidism. This sentence may defend the physician’s medical career by ensuring no patient has an abnormal TSH during thyroid therapy, but it breaks the medical oath to “do no harm.”
Scientific and medical blindness leads to a failure in medical ethics, human suffering, and higher medical costs, but thyroid medical dogma must be defended or the physician’s career is in jeopardy. It’s not just a system that monitors a patient’s therapy. It’s a system that monitors physician compliance and emasculates medical discernment.
Now let’s find a way out for the patient trapped in this dead-end situation.
How to diagnose central hypothyroidism during thyroid therapy
If TSH secretion is abnormally sluggish, neither the physician nor the patient can accuse TSH and win their case unless they have laboratory evidence and can back up an argument with science.
This article will provide you with the tools to prove (or disprove) the case of a sluggish TSH secretion rate.
- Know how to bypass lab “TSH-reflex” flowcharts that force laboratories to cancel FT3 and FT4 lab tests against the physician’s orders. Without laboratory evidence, no one can build a case that proves TSH misbehavior.
- Know what central hypothyroidism looks like during standard levothyroxine (LT4) thyroid therapy. It will look different from central hypothyroidism pre-therapy, and different from primary hypothyroid patients with normal TSH responses. Graphs are provided.
- Use the free SPINA-Thyr endocrinology research tool to analyze TSH, FT4 and FT3 lab results when a patient is on LT4 therapy. The “TSHI” (TSH index) has been developed by scientists and intended for use in detecting pituitary failure even during LT4 therapy. This tool reveals statistical abnormalities in TSH-FT4-FT3 hormone relationships, and an example is provided.
- Use Hadlow’s scientific graph of normal TSH-FT4 relationships in 120,000+ healthy adults. Learn how to visually “map” or plot the patient’s relationships onto this graph while accounting for the different lab reference ranges. Understand how age and sex affects the normal TSH-FT4 relationship within the two hormones’ normal reference ranges.
- Follow through the rest of the diagnostic flowchart for central hypothyroidism, because the TSH is often not the only pituitary hormone that fails to secrete appropriately. Other pituitary or hypothalamic hormone deficiencies, such as growth hormone or cortisol deficiency, or problems with vasopressin (antidiuretic hormone, ADH), must be treated if they exist (Benvega et al, 2018). Other family members may have similar genes that contribute to a pituitary handicap.
Obtain TSH, FT3 and FT4 Bypass the TSH-reflex test cancellation system
One can’t build a case without evidence.
TSH and Free T4 are necessary as a bare minimum for central hypothyroidism diagnosis.
But when the case is unclear because either TSH or FT4 is normal, the Free T3 is necessary to aid diagnosis. This is because a higher FT3 can explain why TSH is normal when it should be high, or why the TSH is low when it should be normal. In contrast, a lower FT3 than expected can strengthen the case for central hypothyroidism.
A barrier to obtaining FT3 and FT4 data: the TSH-reflex system
Beck-Peccoz and colleagues have made it very clear that the “TSH-reflex strategy” works against the diagnosis of central hypothyroidism.
“An increasing number of laboratories use the TSH-reflex approach for screening thyroid function as a first-line test for measuring levels of TSH. It would be impossible to identify patients with central hypothyroidism using this method.”
(Beck-Peccoz et al, 2017)
Unfortunately, in many jurisdictions, a normal TSH may result in cancellation of FT3 and FT4 tests.
But if a suspected central hypo patient is on thyroid hormone therapy, their TSH may be falsely normalized by underdose. The only way to prove their normal TSH is hiding an underdose is to measure FT3 and FT4 and discover that they are inconsistent with their TSH.
Laboratories take orders from health care administrators. Many of them have been persuaded by penny-pinching “Choosing Wisely” campaigns, and the misguided endocrinologists who support them, to say that FT3 and FT4 tests are “not indicated” whenever TSH is normal. Laboratories need to prove to such administrators that they have a medical basis to put aside the flowchart and test FT3 and FT4 anyway.
A physician can give the laboratory the freedom to test FT3 and FT4 by using the correct code words on the requisition. The doctor may need to write in the explanation box “suspected central hypothyroidism. Do not cancel FT3 or FT4.”
Some laboratory staff members would love to have the opportunity to be medical heroes. They don’t want to be mindless lackeys. It was a laboratory staff member that finally rescued a woman from 23 years of undiagnosed central hypothyroidism due to her inappropriately normal TSH. I have recently seen a notice placed on a suspicious set of lab results saying that the tests were run twice and the results were confirmed. Lab staff want to help, and they can help if we give them the right reasons to disobey anti-diagnostic testing flowcharts.
What CeH looks like during LT4 therapy
Once you have full lab data, here is a pattern you can look for in thyroid lab tests.
During LT4 therapy, the TSH of patients with central hypothyroidism (triangles) is shifted significantly lower than healthy controls (dots) per unit of Free T4, but TSH still responds to FT4.

When a central hypothyroid patient’s FT4 is normalized by thyroid therapy, TSH is usually lower than the median.
(NOTE: Most standard 95% reference ranges are -2 to +2 standard deviations from the “mean” but the TSH has a very skewed population distribution so the median or 50th percentile is often used after log-normalization. This article referred to the “mean,” as shown below in quotations.)
Among the patients with central hypothyroidism,
- “Five patients had baseline TSH level above 4 mU/L, and 9 had pretreatment TSH less than 0.5 mU/L, but only two presented with TSH less than 0.1 mU/L.
- “Pretreatment thyrotropin (TSH) level (mean ± standard error of the mean [SEM]) was 2.04 ± 0.25 mU/L (normal, 0.4–4)”
- TSH “gradually decreased to 0.51 ± 0.19 mU/L (range, 0.009–3.38) by treatment with levothyroxine in a mean dose of 86 ± 6 mcg/d.”
- “TSH was suppressed by thyroid replacement to less than 0.5 mU/L in 80% of patients.”
- “Mean baseline free thyroxine (FT4) was 7.55 ± 0.51 pmol/L (normal, 11.8–24.6) and gradually increased with thyroid hormone to 15.19 ± 1.0 pmol/L.”
- “In the studied patients, TSH level above 1 mU/L was associated with inadequate thyroid hormone replacement and low FT4 in most subjects.”
- “In contrast, TSH below 0.1 mU/L generally predicted euthyroidism in the treated patients” and their belief was that “euthyroidism is usually based on achievement of normal range [Total] T4 levels,” based on a 1999 study.
Sadly, back in 2002, the researchers’ goal was to merely “normalize” FT4 rather than optimize the dose to remove hypothyroid symptoms. Their goal was to prove that the TSH still responded to FT4 in the subclass of central hypothyroidism patients whose TSH is capable of rising into normal range or above. As a result, most patients shown in this graph likely remained underdosed with FT4 in the lower half of range. Their population had an average dose of LT4 at only 86 ± 6 mcg/day.
You might wonder what their T3 was doing. In Shimon’s study, the Total T3 level did not rise from pre-treatment levels during LT4 therapy, and Free T3 was not measured. This was 2002, when good Free T3 tests were hard to come by.
Know what target FT4 (and FT3) ought to be in central hypothyroidism on LT4 therapy
Obviously, TSH normalization is not the target of central hypothyroid therapy because TSH is likely to be suppressed during euthyroid levels of thyroid therapy.
As of 2018, the guidelines for central hypothyroidism in Europe are to achieve a FT4 above the mid-reference range median in untreated populations:
“It is important to evaluate the adequacy of the replacement after 6–8 weeks measuring FT4 and targeting this parameter above the median values of the reference range.”
(Persani et al, 2019, “The diagnosis and management of central hypothyroidism in 2018”)
According to Ganslmeier, 2013, the healthy population’s FT4 median is around 44% of range:

This FT4 treatment target as of 2018 is quite different from what it was 16 years prior, which was often a merely “normal FT4” (as Shimon et al, 2002 believed), or an LT4 dose based on body weight without regard for individual variability in LT4 absorption and metabolism.
Scientists eventually realized that many central hypothyroid patients were undertreated. The shocking thing is how long it took for them to realize this.
A major step forward was Kuolouri et al’s 2011 study that compared central hypothyroidism on LT4 with primary hypothyroidism on LT4, rather than comparing their FT4 levels with the population distribution found in untreated patients:
“the distribution of fT4 in patients on levothyroxine needs to be higher than the normal population in order to maintain a euthyroid state as defined by normal TSH levels.
Therefore we suggest that clinicians in our department may be accepting [lower normal] fT4 levels which they are familiar with in euthyroid individuals as adequate replacement, whereas in fact these levels are probably suboptimal.”
(Koulouri et al, 2011)
Alas, the misguided thyroid treatment philosophy of biochemical mimicry is at the foundation of so many thyroid treatment failures!
Why must treatment mimic the biochemistry of healthy populations who don’t have a thyroid or pituitary disorder and aren’t on thyroid medication? Can’t reasonable people admit that an HPT axis gland’s failure and its treatment may fundamentally distort the HPT axis, making a low-normal FT4 unacceptable without a higher FT3 to compensate? Therapeutic intervention and compensation is also a valid treatment philosophy.
But what’s the upper limit? How much thyroid hormone is too much? How high do levels need to be for “overtreatment” to exist in central hypothyroidism?
This is where FT3 and symptoms come into the picture:
“An excessive LT4 intake should be considered whenever FT4 concentrations are above or at the upper limit of the normal range, in particular when clinical manifestations of thyrotoxicosis and/or high FT3 levels are present.”
(Persani et al, 2019)
Use SPINA-Thyr to quantify TSH misbehavior
While a patient is only dosing levothyroxine (LT4) and no T3 or desiccated thyroid hormone, use the free endocrinologist SPINA-Thyr program to give a more careful mathematical analysis of pituitary gland health given TSH, Free T4, and Free T3 levels.
In particular, the TSHI, or “TSH index” is an index of TSH response to FT4 and FT3 developed by Jostel and team in 2009 based on analysis of 9,519 tests in 4,064 adults. As explained by the creators of this index,
“The TSHI is a numerical estimate of the pituitary thyrotroph function that can be derived from simple thyroid function tests, allowing easier detection of pathological TSH suppression in hypopituitarism regardless of concomitant T4 supplementation.
(Jostel et al, 2009)
Jostel’s TSHI was adopted by SPINA-Thyr, developed by research endocrinologist Johannes W. Dietrich. The research tool has been thoroughly tested and reveals findings of significance in many population, including LT4-treated patients (Dietrich et al, 2016). The tool continues to be used by research scientists in recent publications.
First, I’ll provide a case study and show what can be seen by analyzing the results without SPINA, then how the results are further analyzed with SPINA.
A case study of central hypo lab results on LT4 therapy
Here is an example of a case of strong suspicion for central hypothyroidism in a real patient who was diagnosed with primary hypothyroidism.
This patient was dosing 112.5 mcg LT4 plus 10-15 mcg/day T3. She stopped dosing her T3 for 12 days prior to testing for the sake of finding out what her GD and pituitary function would be.
Twelve days of T3 withdrawal was more than sufficient, since the half-life of T3 is no more than 2 days. LT3 withdrawal studies in research show that it only takes 5 days after T3 withdrawal to remove the effect of T3 dosing on FT3 levels. On an earlier, less sensitive TSH assay, it took at least 10 days to achieve “high” TSH in athyreotic thyroid cancer patients who had maintained a suppressed TSH for one month on T3 monotherapy prior to complete withdrawal of T3 (Hilts et al, 1979).
She suffered from this experiment with hypothyroid symptoms, but she wanted to obtain data that could be useful in diagnosis.
Test 1.
- LT4 dose: 112.5 mcg/day
- TSH 0.23 (0.32 – 4.00)
- FT4 13 (9.0 – 19.0) = 40% of the reference range width
- FT3 2.7 (2.6 – 5.8) = 3% of the reference range
- RT3 14.0 (8.0 – 25.0) = 35.3%
- FT3:FT4 ratio: 0.20 (poor converter on LT4 is <0.25 according to Midgley et al, 2015))
Test 2.
It’s important to have more than one test result to ensure the TSH hyposecretion is not transient or “on the move.”
Fifteen days later, the tests were repeated at the same dose of LT4, with the following findings not much different.
- LT4 dose: 112.5 mcg/day
- TSH 0.19 (0.32 – 4.00)
- FT4 14 (9.0 – 19.0) = 50% of the reference range width
- FT3 3.2 (2.6 – 5.8) = 19% of the reference range
Her FT4 levels do not meet the criteria for euthyroid levothyroxine therapy in a patient with central hypothyroidism because the FT4 is not above the healthy population mean for FT4 (Persani et al, 2019), which is approximately 45% of reference (Ganslmeier et al, 2013) and she was symptomatically hypothyroid.
Her FT4 levels do not meet the criteria for overtreatment on levothyroxine therapy in a patient with central hypothyroidism (provided above) because the FT4 is not above the reference range, and neither is the FT3.
However, without SPINA-Thyr to show the insufficiency of her TSH response to her “normal” FT4 and FT3, this patient would have her dose reduced as punishment for the sins of her pituitary gland.
SPINA-Thyr results for lab test #1
Lab result set #1 revealed the following evidence of metabolic failure and pituitary failure:
- GD: 19.20 nmol/s (20 – 40). This “global deiodinase efficiency” index is derived from the FT3:FT4 ratio, which is a measure of net peripheral T4 to T3 metabolism. It is common for people with very little residual thyroid gland function to have a low-normal GD and low FT3:FT4 ratio while on levothyroxine (Midgley et al, 2015). This points to either LT4-treated severe primary hypothyroidism with poor conversion, or severe nonthyroidal illness. However, the RT3 result shown above, in comparison with the FT4 result excludes nonthyroidal illness, leaving primary hypothyroidism.
- sGD: -2.16 (-2.0 to +2.0). This is the standard deviation from the population mean of zero (0). Most 95% laboratory reference intervals cover 2 standard deviations on both sides of the mean. This result is far below the range found in healthy people.
- TSHI 0.3 (1.3 to 4.1). This is an index of pituitary TSH concentrations expected in response to a patient’s current FT4 and FT3 levels. The result is far below range.
- sTSHI -3.58 (-2.0 to +2.0). This is the standard deviation from the population mean of zero (0). This patient’s pituitary TSH secretion rate is inappropriately low given her thyroid hormone levels.
- TTSI 16 (100-150). This is the TSH response to FT4 alone, in relation to the top of the FT4 reference range. Its use is mainly for the detection of the opposite condition, the inappropriate hypersecretion of TSH when FT4 is high-normal or high. TTSI is expected to be low when the TSHI is low.
See our free walkthrough on SPINA-Thyr.
Map your lab results onto the normal TSH-FT4 relationship
If you have enough lab history without T3 dosing, you can plot a graph with data. You should be able to see a pattern, as long as you also know what the “normal” TSH-FT4 relationship looks like.
To a person who lives near the Rocky Mountains, it looks a little bit like a ski hill.
The “inappropriate” TSH per unit of FT4 can be seen against this background.

CAUTION: The graph can be misread if you don’t look closely at the labeling of the axes.
Notice the X axis for Free T4 is linear, adding +5 each time: 5, 10, 15, 20, 25 pmol/L.But the TSH Y axis tickmarks are at multiples of x10 : 0.1, 1, 10, 100, 1000. Without doing this distortion, the graph would be extremely tall.
This Y axis is not a visual distortion. The natural relationship between TSH and FT4 is “log-linear,” meaning TSH response is logarithmic in response to a linear change in FT4.
It is physiologically appropriate to expand the region under the “ski hill” of normal data with the logarithmic scale, because below reference range, it takes more and more FT4 to push the TSH down by 0.1.
As you can see, the “normal” TSH-FT4 log-linear relationship takes a wobbly diagonal journey through part of the normal reference range. It does not cover the whole overlapping reference range, the dotted-line rectangle in the middle of the graph.
The basis of the graph
The graph above represents data from 120,403 people, which is a huge number.
They EXCLUDED patients with central hypothyroidism, as well as other categories of people who could have had abnormal TSH secretion for various reasons:
“We excluded 79 719 records (15.2%)” who were from “hospitalized patients, pregnant women, patients aged younger than 1 year, patients receiving specialist endocrine, surgical, or medical care . . . treated Graves disease or thyrotoxicosis, multinodular goiter, thyroid cancer, partial or total thyroidectomy, and hypopituitarism” and “and those with a history of treatment with radioiodine, antithyroid drugs, lithium, antiepileptic drugs, amiodarone, or liothyronine [T3].”
(Hadlow et al, 2013)
They analyzed the TSH-FT4 relationships of LT4-treated patients separately, but I don’t recommend using that graph. It’s very similar to the untreated population’s “ski hill” anyway. It’s not wise to use it because it is tainted by non-optimized thyroid therapy by poorly educated physicians who only know how to normalize, not optimize. That data set may may include patients with central hypothyroidism who have not been diagnosed.
Know where the “central hypothyroidism” zone is on the graph
The zone is under the “ski hill” that describes the 95% population reference interval for TSH at every level of FT4.

The blue zone is constructed based on research cited below the graph and in this article.
Notice that part of the “central hypo zone” is within the lower corner of the two hormones’ overlapping reference ranges. Naive doctors are trying to force hypopituitary patients into that zone. We really do need graphs to see how our hormones are responding inappropriately to each other.
Some people may wonder why the TSH is mildly high on the far left side of the “central hypo zone.” This is also a region for central hypothyroidism, as revealed by Beck-Peccoz et al, 2017.
The mildly high TSH zone is where central and primary hypothyroidism overlap and can masquerade as each other. Hypothalamic or “tertiary” central hypothyroidism can present with mildly elevated TSH, as shown in Beck-Peccoz’s 2017 graph above. “Tertiary hypothyroidism can be misdiagnosed as a condition of primary hypothyroidism” (Beck-Peccoz et al, 2017), either overt with low FT4, or subclinical with normal FT4. In fact, the patient may have central hypothyroidism, or both primary and central forms of thyroid disease.
Finally, part of the “central hypo zone” occurs underneath high-normal FT4 levels. In a 2020 article by Boronat and colleagues, this potential of overlooking central hypothyroidism as the opposite condition of hypER was explained. The article’s title was “Central hypothyroidism or subclinical hyperthyroidism: can they be confused with each other?” The answer is, YES.
“In these cases, diagnosis is challenging, as symptoms are unspecific and usually mild, and laboratory findings are variable, including low, normal or even slightly elevated TSH levels, along with low or low-normal concentrations of free T4.”
(Boronat et al, 2020)
Another graph from Shimon et al, 2002, includes TSH-FT4 relationships before and during LT4 therapy:

As shown in the graph above,
- TSH will often fall low before FT4 rises to mid-range in central hypothyroidism.
- TSH begins to rise only as FT4 falls into the lower quarter of range during LT4 therapy
- TSH may be normal only very close to the bottom of the FT4 range and below it.
If a doctor wanted to keep a patient’s TSH normal, you can see where they will put the patient’s FT4, too low.
It is a mistake to presume that the TSH is the infallible “thyroid thermostat” that “knows best” where the patient’s FT4 should be, when this is where the pituitary-disabled patient’s “normal” TSH steers the FT4 to be — far too low for most people in the healthy population.
Next, use a graphics program to map your TSH-FT4 relationships on the graph
- Re-label the X and Y axis labels so that the “normal range” square in the middle lines line up with YOUR lab reference ranges. (Each lab will have a slightly different range that fits their lab test manufacturer and population.) Here, the TSH reference range is (0.4–4.0 mU/L) and free T4 reference range is (10–20 pmol/L). For example, if your Free T4 reference range is 10 to 25, cross out their “20” and replace it with “25.” You can do the math to fix the other tickmarks so that the same number of units is between every two tickmarks.
- Find your Free T4 level on the corrected X axis. Follow a line with your eyes to see where the average (triangle) and range (dotted lines) of expected TSH values would be at the given FT4 level.
This graph shows Lab results from Test 1 and Test 2 in the case study above:
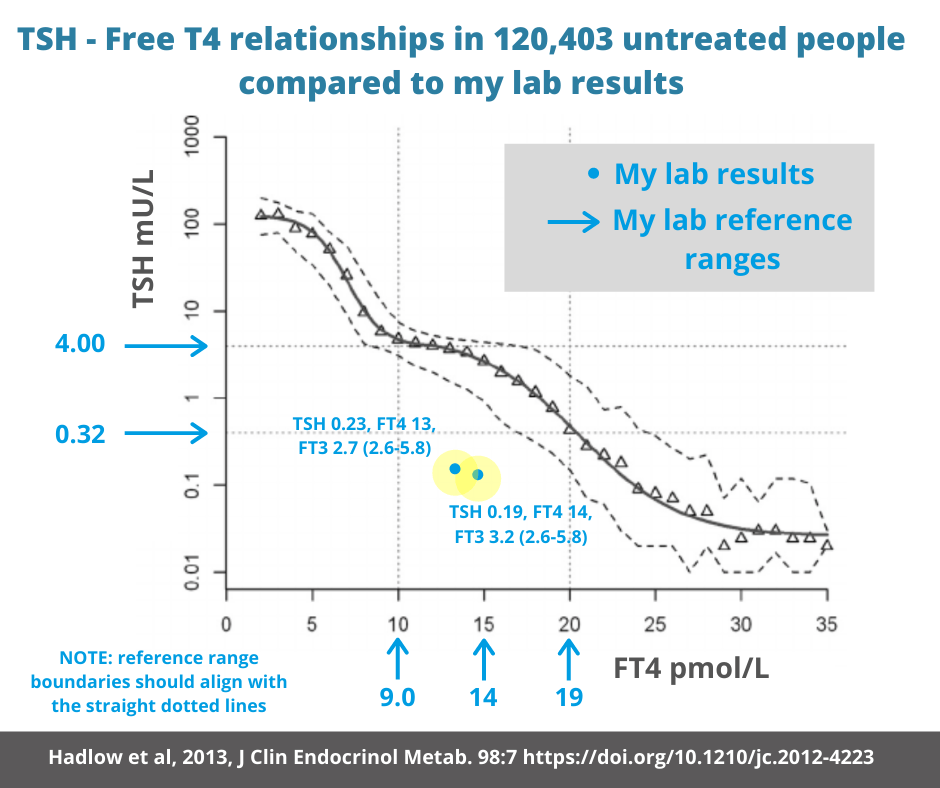
The blue dots stand out as clear cases of central hypothyroidism despite being normal FT4 values.
The blue dots do not fall into the usual case of subclinical hyperthyroidism which is within the “ski hill’s” dotted lines. In most cases of true subclinical hyper (with or without central hypothyroidism), TSH falls mildly below its range just before FT4 rises above its range. This is often because the FT3 is elevated in isolation by mild Graves’ disease or a T3-secreting thyroid nodule.
Next, are you male or female?
The next two graphs by Hadlow et al, 2013 zoom into the reference range. They also become linear, not logarithmic, on their Y axis for TSH.
There is insufficient room for the blue dots underneath the dotted line because the huge region under the TSH reference range and above complete suppression is collapsed by the linear scale.

You can see that the sex difference matters if you are in the normal range for FT4 and TSH during thyroid therapy but you fall into the lower left corner of the so-called “euthyroid” rectangle, which is actually part of the “central hypo zone.”
Next, how old are you?

As shown, if one is under 39 years old, the TSH-FT4 relationship is very different from that of older people. As a person ages, the region of central hypothyroidism expands under the curve, as TSH rises, rather than falls, when FT4 is normal in untreated adults.
Something is very wrong with pituitary TSH secretion if an older person has a mid-range FT4 and mid- to low-range FT3 and a very low TSH.
If more physicians were to study these graphs closely, and were to ponder their implications for pituitary TSH secretion inhibition, it ought to be easier to diagnose central hypothyroidism in people of different ages.
The diagnosis of central hypo
Let’s assume you have completed a SPINA-Thyr analysis and/or Hadlow graph plotting exercise, and it has verified a TSH that is lower than it should be for your FT4 and FT3, sex, and age. Here are the next steps in diagnosis.
The 2018 diagnostic flowchart for central hypothyroidism
If you can’t zoom in, see the original article online.

The top of the flow chart specifies “Low or low-normal FT4 and inappropriately low TSH.” But the words “low” and “low normal” can be deceptive. The TSH reference range boundaries are not diagnostic barriers, as shown above — it’s the “inappropriate” relationship between the hormones that matters. An “inappropriately low TSH” can include an “inappropriately normal TSH” when it ought to be an elevated TSH, or a mildly elevated TSH that ought to be a significantly elevated TSH.
To go through the flowchart, you will need to know more about the differential diagnoses and potential causes and whether they line up with anything in your medical history.
Consider differential diagnoses
Certain syndromes can mimic the biochemistry of central hypo, especially when TSH and FT4 are viewed in isolation from FT3 and other clinical factors.

The list of other conditions does not mean that CeH is absent when they are present. It means that these conditions may be “transient,” or that a damaged or dysfunctional pituitary TSH secretion may be confused with these conditions because they share some characteristics.
The RT3 and FT4 relationship can detect “nonthyroidal illness” as a cause of pituitary TSH inhibition
As mentioned at the top of the list in Persani’s table above is “Severe form of nonthyroidal illness or sick euthyroid syndrome.” Illnesses can falsely keep TSH normal or push the TSH secretion rate lower as T3, and then T4, falls over time. Some scientists admit that this is a form of central hypothyroidism (Teblick et al, 2019).
TSH-distorting illness can be ruled in or out by comparing Reverse T3 (rT3) with Free T4 (FT4) in terms of their relative positions in or around their reference ranges.
Nonthyroidal illness syndrome (NTIS) elevates the RT3 level in relationship to FT4 due to a significantly higher RT3 appearance rate (via upregulated D3 enzyme) and slower RT3 clearance rate (via downregulated D1 enzyme).
When NTIS is present, if RT3 may be 50% of range higher than the FT4 is within their respective reference ranges.
What causes this? The distortion of all three enzymes is induced by tissue hypoxia and/or inflammatory cytokines such as IL-6. Therefore, it is no surprise that even in “low-grade systemic inflammation,” researchers have found “Inflammation predicted the FT4/rT3 ratio” (Moura Neto et al, 2013).
Using Test #1 data in the case study above, the hormone relationship rules out the effect of nonthyroidal illness syndrome (NTIS) as a possible reason for the lower FT3:FT4 ratio and lower TSH.
The Reverse T3 test result in relation to the FT4, its only hormone of origin, are a close fit in terms of their relative percentage of the reference range: 35.3% of reference for RT3 and 40% of reference for FT4.
There is no distortion in the RT3-FT4 relationship in the patient, so we cannot blame NTIS. This fits the biochemical pattern of health during LT4 monotherapy. In health, FT4 and RT3 have a strong correlation and rise in parallel, and FT3 also rises per unit of FT4 (Massolt et al, 2016).
However, even if the RT3 was significantly elevated above the FT4’s position of range, illness can occur within central hypothyroidism, so one must not treat them as mutually exclusive conditions!
Drugs that can oppress TSH
Drugs that inhibit TSH secretion can be learned by scanning articles such as Haugen et al, 2009, “Drugs that suppress TSH or cause central hypothyroidism,” or the more up-to-date chapter by Thalmann & Meier, 2013, and they include:
- Dopamine/Dopamine Agonist
- Somatostatin Analogs
- Rexinoids
- “Antiepileptic drugs such as carbamezipine, oxcaremazepine, valproic acid [valproate], and phenytoin”
- “Metformin, the first-line recommended antidiabetic agent in type 2 diabetes, reduces TSH levels in patients with hypothyroidism (treated or not) without altering free T4 levels, but not in diabetics without concomitant hypothyroidism.”
Additional reasons
It’s easy to rule in or out pregnancy or neonatal effects on TSH.
A major condition not listed by Persani above is T3 therapy or desiccated thyroid therapy, which can suppress or lower TSH at euthyroid doses even when FT4 and FT3 are both in reference range. There are many reasons for this. It has long been known that even metabolically euthyroid oral T3 dosing can be a TSH suppressant. It is largely due to the benign fluctuations in Free T3 during dosing.
In addition, if you suspect that you were misdiagnosed with primary hypothyroidism and your thyroid gland is actually functional, you could try assessing your thyroid gland health and antibody status:
- Thyroid ultrasound. If the thyroid is completely normal and no signs of damage are present, then it’s possible the diagnosis of primary hypothyroidism was a mistake made when TSH was mildly inflated, which can happen in central hypothyroidism (see above).
- Antibody testing of thyroid peroxidase antibody (TPOab) and thyroglobulin antibody (TGab) to rule out Hashimoto’s thyroiditis. Ruling out Hashimoto’s makes central hypo more likely, because what else would explain hypothyroidism if the most common cause is not present? However, TPOab positivity does not rule out central hypo, because patients with Hashimoto’s can have central hypo, as well.
Persani mentions in his list “Thyrotoxicosis-related conditions: prolonged TSH suppression after recovery from thyrotoxicosis.” This “prolonged suppression” is actually caused by antibodies. Recovery from thyrotoxicosis is biochemical. It does not always coincide with remission from TSH-receptor autoimmunity. These antibodies may relapse later even if there is a remission.
- If the patient has autoimmune thyroid disease, test for Graves’ disease antibodies — TSH receptor antibodies (TRAb test). One type of TRAb, the TSH receptor stimulating antibody (TSAb), can artificially reduce TSH by interfering with the pituitary “ultrashort feedback loop” even when the thyroid is disabled and thyroid hormones are normal. (See “The TSAb stimulating antibody can lower TSH despite euthyroid status.“)
- TSAb are present in up to 10% of patients with Hashimoto’s thyroiditis (See “Overlooked: How many Hashimoto’s patients with TSH-Receptor antibodies?“).
- TSAb antibodies are even more likely if a patient has Atrophic Thyroiditis (very small thyroid gland volume, irregular shape, fibrosis, low echogenicity), because autoimmune thyroid atrophy is genetically aligned with Graves’ disease and caused by the TSH Receptor blocking antibody (Jara et al, 2009).
Consider acquired CeH causes
Have you ever had a concussion or hit your head very hard? Traumatic brain injury is a major cause of CeH.
Consider the following in light of your health history:

The table on “congenital” and genetic causes is too large to duplicate here and would have to be viewed at the source: see the original article online.
Conclusion
The challenges of diagnosis of central hypothyroidism during thyroid hormone therapy are caused by excessive medical trust in TSH normalization as a therapeutic target. This use of TSH has blinded physicians from diagnosing abnormal thyroid hormone – TSH relationships during thyroid therapy.
Physicians ought to be looking out for TSH misbehavior, not defending the practice of underdosing levothyroxine to normalize a misbehaving TSH.
Medical systems don’t change just because individuals fall through the cracks. They only change when large groups of people fall through the cracks and cost the health care system money, or when the identification of such groups provides a new opportunity for pharmaceutical profit or scientific credit.
I believe systemic change will only come after researchers provide large scale data regarding medical errors. Until researchers publish their work, the discovery of central hypothyroidism during thyroid therapy is likely going to happen as a result of self-education by patients and physicians.
Science-based patient organizations like ours are offering compassionate doctors the evidence and scientific tools they need to defend their patients and their medical careers at the same time.
We know our doctors will need a strong shield to defend themselves. They will need to use science and critical thinking to save a “rare” (or not so rare?) patient whose pituitary gland can’t obey the HPT axis rules.
I believe that good science and good clinical evidence are the shield and sword of the misunderstood thyroid patient and their evidence-based physician. We patients and physicians must equip each other with science and critical thinking, educate each other, stand together, and defend each other.
The diagnosis of central hypo during thyroid therapy is complex and challenging, but it is possible with enough scientific knowledge and relevant health data.
Best wishes,
Tania Sona Smith, PhD
President, Thyroid Patients Canada,
Thyroid patient and thyroid science analyst
References
View reference list
Beck-Peccoz, P., Rodari, G., Giavoli, C., & Lania, A. (2017). Central hypothyroidism—A neglected thyroid disorder. Nature Reviews. Endocrinology, 13(10), 588–598. https://doi.org/10.1038/nrendo.2017.47
Benvenga, S., Klose, M., Vita, R., & Feldt-Rasmussen, U. (2018). Less known aspects of central hypothyroidism: Part 1 – Acquired etiologies. Journal of Clinical & Translational Endocrinology, 14, 25–33. https://doi.org/10.1016/j.jcte.2018.09.003
Boronat, M. (2020). Central hypothyroidism or subclinical hyperthyroidism: Can they be confused with each other? Endocrinology, Diabetes & Metabolism Case Reports, 2020(1). https://doi.org/10.1530/EDM-20-0059
Dietrich, J. W., Landgrafe-Mende, G., Wiora, E., Chatzitomaris, A., Klein, H. H., Midgley, J. E. M., & Hoermann, R. (2016). Calculated Parameters of Thyroid Homeostasis: Emerging Tools for Differential Diagnosis and Clinical Research. Frontiers in Endocrinology, 7. https://doi.org/10.3389/fendo.2016.00057
Ganslmeier, M., Castrop, C., Scheidhauer, K., Rondak, I.-C., & Luppa, P. B. (2014). Regional adjustment of thyroid hormone reference intervals. Journal of Laboratory Medicine, 38(5), 281–287. https://doi.org/10.1515/labmed-2014-0008
Haugen, B. R. (2009). Drugs that suppress TSH or cause central hypothyroidism. Best Practice & Research. Clinical Endocrinology & Metabolism, 23(6), 793–800. https://doi.org/10.1016/j.beem.2009.08.003
Hilts, S. V., Hellman, D., Anderson, J., Woolfenden, J., Van Antwerp, J., & Patton, D. (1979). Serial TSH determination after T3 withdrawal or thyroidectomy in the therapy of thyroid carcinoma. Journal of Nuclear Medicine: Official Publication, Society of Nuclear Medicine, 20(9), 928–932. https://pubmed.ncbi.nlm.nih.gov/536837/
Jara, L. J., Vera-Lastra, O., & Medina, G. (2008). Atrophic Thyroiditis. In Diagnostic Criteria in Autoimmune Diseases (pp. 221–225). Humana Press. https://doi.org/10.1007/978-1-60327-285-8_42
Jonklaas, J., Bianco, A. C., Bauer, A. J., Burman, K. D., Cappola, A. R., Celi, F. S., Cooper, D. S., Kim, B. W., Peeters, R. P., Rosenthal, M. S., & Sawka, A. M. (2014). Guidelines for the Treatment of Hypothyroidism: Prepared by the American Thyroid Association Task Force on Thyroid Hormone Replacement. Thyroid, 24(12), 1670–1751. https://doi.org/10.1089/thy.2014.0028
Koulouri, O., Auldin, M. A., Agarwal, R., Kieffer, V., Robertson, C., Falconer Smith, J., Levy, M. J., & Howlett, T. A. (2011). Diagnosis and treatment of hypothyroidism in TSH deficiency compared to primary thyroid disease: Pituitary patients are at risk of under-replacement with levothyroxine. Clinical Endocrinology, 74(6), 744–749. https://doi.org/10.1111/j.1365-2265.2011.03984.x
Massolt, E. T., van der Windt, M., Korevaar, T. I. M., Kam, B. L. R., Burger, J. W., Franssen, G. J. H., Lehmphul, I., Köhrle, J., Visser, W. E., & Peeters, R. P. (2016). Thyroid hormone and its metabolites in relation to quality of life in patients treated for differentiated thyroid cancer. Clinical Endocrinology, 85(5), 781–788. https://doi.org/10.1111/cen.13101
Midgley, J. E. M., Larisch, R., Dietrich, J. W., & Hoermann, R. (2015). Variation in the biochemical response to l-thyroxine therapy and relationship with peripheral thyroid hormone conversion efficiency. Endocrine Connections, 4(4), 196–205. https://doi.org/10.1530/EC-15-0056
Ministry of Health, British Columbia. (2018, October 24). Thyroid Function Testing in the Diagnosis and Monitoring of Thyroid Function Disorder—Province of British Columbia. Province of British Columbia. https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/thyroid-testing
Moura Neto, A., Parisi, M. C. R., Tambascia, M. A., Alegre, S. M., Pavin, E. J., & Zantut-Wittmann, D. E. (2013). The influence of body mass index and low-grade systemic inflammation on thyroid hormone abnormalities in patients with type 2 diabetes. Endocrine Journal, 60(7), 877–884. https://doi.org/10.1507/endocrj.ej13-0030
Persani, L., Brabant, G., Dattani, M., Bonomi, M., Feldt-Rasmussen, U., Fliers, E., Gruters, A., Maiter, D., Schoenmakers, N., & van Trotsenburg, A. S. P. (2018). 2018 European Thyroid Association (ETA) Guidelines on the Diagnosis and Management of Central Hypothyroidism. European Thyroid Journal, 7(5), 225–237. https://doi.org/10.1159/000491388
Persani, L., Cangiano, B., & Bonomi, M. (2019). The diagnosis and management of central hypothyroidism in 2018. Endocrine Connections. https://doi.org/10.1530/EC-18-0515
Shimon, I., Cohen, O., Lubetsky, A., & Olchovsky, D. (2002). Thyrotropin suppression by thyroid hormone replacement is correlated with thyroxine level normalization in central hypothyroidism. Thyroid: Official Journal of the American Thyroid Association, 12(9), 823–827. https://doi.org/10.1089/105072502760339406
Téblick, A., Langouche, L., & Van den Berghe, G. (2019). Anterior pituitary function in critical illness. Endocrine Connections, 8(8), R131–R143. https://doi.org/10.1530/EC-19-0318
Thalmann, S., & Meier, C. A. (2013). Effects of drugs on TSH secretion, thyroid hormones absorption, synthesis, metabolism, and action. In L. E. Braverman & D. S. Cooper (Eds.), Werner & Ingbar’s the thyroid: A fundamental and clinical text (10th ed..). Wolters Kluwer/Lippincott Williams & Wilkins Health.

Leave a public reply here, on our website.