
A normal ratio of Free T3 (FT3) to Free T4 (FT4) thyroid hormones appears to be a basic principle in thyroid hormone economy when no thyroid hormone dosing and no disease interfere.
In this post, I provide graphs, quotations, and discussions from Gullo, Strich, and Anderson as I present their FT3:FT4 ratio results, and I rely on science to present the mechanisms by which the ratio is adjusted in relationship to TSH in people not dosing thyroid hormones.
Comparing across these 3 different studies, you’ll see how amazingly consistent population average FT3:FT4 ratios are among people with healthy thyroid glands in various age and sex categories. There seems to be a standard ratio in blood while TSH is normal:
- The healthy average is approximately 0.31 to 0.34 mol/mol.
This is because the healthy thyroid, healthy pituitary, and healthy metabolism stabilize the balance of FT3 per unit of FT4 in blood.
Keep in mind, however, that these data tend to look more stable at the population level — it conceals some diversity between individuals.
Technical basics
How is the ratio calculated? One can only create a free hormone ratio when measuring both as Free (not Total T3 to Free T4).
Both must be based on FT3 and FT4 from the same blood draw.
Both ought to be measured at a single laboratory, using a standard immunoassay for FT3 and FT4 on a single platform (i.e. Siemens, Roche, Abbott, and so on).
To make a ratio within this system,
- First, the lab results must be expressed in pmol/L. American units are in weight per volume, but pmol/L is a chemical unit of measure, based on the size of the molecule. If working with American units, The American Medical Association (AMA) conversion calculator provides the conversion factors as
- FT4 ng/dL x 12.871 = result in pmol/L,
- FT3 pg/mL x 0.0154 = result in pmol/L.
- Divide the individual’s FT3 by their FT4. (5.0 pmol/L FT3 divided by 15.0 pmol/L FT4 is a ratio of 0.33 mol/mol).
Most of these studies use pmol/L. Although many research articles have used ng/dL and other units, I prefer studies that use molar units (pmol/L) for the individual blood draws.
Anderson’s data set below was measured in American units, but it is included for its rich and unique data set. It was converted to pmol/L before dividing the average FT3 by the FT4. This enabled comparison to Gullo’s and Strich’s results in pmol/L.
However, this is not ideal. Converting a population’s average ratio in ng/dL to pmol/L (to obtain a ratio of averages) is not the same as obtaining ratios in pmol/L from each individual (an average of ratios).
FT3:FT4 ratios must always be anchored in absolute blood levels. They are an assessment of global metabolic rate, not systemic hypo- or hyperthyroid status.
This example demonstrates how the same ratio may induce hypo or hyper:
- A high ratio such as 0.46 may be seen in people with untreated early Hashimoto’s hypothyroidism before treatment, when FT4 drops below range and their high TSH is stimulating the functional thyroid tissue that remains.
- TSH receptor stimulation preferentially enhances the T3 side of the T4:T3 ratio of secretion from thyroid tissue.
- D2 enzyme in peripheral tissues will be upregulated when FT4 is low, enhancing peripheral T4-T3 conversion rate.
- The same ratio of 0.46 may be seen in a person with untreated early Graves’ hyperthyroidism in a hypothetical person with a FT4 near the top of reference range, a high FT3, and 0.001 TSH.
- This ratio is expected because instead of TSH hormone, TSH-receptor stimulating antibodies are hijacking TSH receptors and overstimulating the thyroid.
- The ratio may reduce and wobble as hyperthyroidism becomes more severe and FT4 increases, because these antibodies can fluctuate wildly, and hyperthyroidism upregulates D1 and D3 enzymes at different rates in different tissues.
Caution. These ratios don’t apply during thyroid hormone treatment.
Just as the “normal” ratio “in large populations” is not the prescription for the healthy individual within that population, the “normal ratio” is especially not the prescription for the thyroid-disabled, pharmaceutically-supplied individual.
The ultimate goal of thyroid therapy is not to achieve a biochemical target established by another population’s statistical norms. Instead, the goal is to yield appropriate levels of T3 signaling in receptors within an uniquely disabled and dose-manipulated metabolic system.
During hormone treatment, a healthy, optimal FT3:FT4 ratio in blood is individualized to a person’s unique metabolic handicaps and demands.
If you want FT3:FT4 ratio statistics for diverse hypothyroid populations on LT4 monotherapy, see our other articles, such as “Are you a poor T4 converter? How low is your Free T3?.”
As for ratios in people dosing T3 hormone, we don’t have enough published population data yet to create benchmarks for FT3:FT4 ratios in desiccated thyroid or specific stable T4:T3 dosing ratios. Dose ratios and even “number of hours since dose” will inevitably influence the data. Many clinical trials of T3-inclusive therapy have measured Total T3, not Free T3. None so far have collected FT3:FT4 ratios for every individual. We need more research, and better research.
One should neither expect nor force a thyroid-disabled, hormone-dosing person to imitate the healthy average ratio of the undosed healthy population. Thyroid disease and dosing introduce significant shifts in oral hormone absorption and metabolism. The FT3:FT4 ratio and levels in blood that used to support health pre-thyroidectomy might no longer support health post-thyroidectomy. This is mainly because of 3 reasons:
- A TSH-regulated thyroid gland compensates for — and conceals — underlying metabolic handicaps in T4-T3 conversion in tissues beyond the thyroid gland. Loss of thyroid function gradually or suddenly exposes these handicaps, which differ from person to person. See “Thyroid T3 secretion compensates for peripheral T4-T3 conversion.”
- GI tract absorption of hormones in pulses is very different from thyroidal secretion of hormone directly into bloodstream at a gentle TSH-regulated sine wave of circadian rhythm.
- In people who dose T3 and/or T4, some hormone is influencing, and being influenced by, the GI tract and microbiome even before it enters systemic circulation. Researchers have only begun to study this dynamic.
- LT4 monotherapy creates post-dose FT4 peaks. In people without thyroid function, LT4 monotherapy obliterates the natural FT3 circadian rhythm that enables healthy people’s FT3 to rise higher at night, while sleeping.
- LT3 dosing creates significant, yet predictable post-dose FT3 peaks and valleys. Therefore, LT3 may be dosed and timed to imperfectly imitate circadian rhythms. The body compensates for FT3 peaks and valleys by continually shifting the metabolic rate of both T3 and T4 hormone throughout the day, as D1 and D3 enzymes are powerfully upregulated by T3 signaling.
Due to these factors, pharmaceutical dose ratios (micrograms of LT4 to LT3 dosed) will never reliably produce the same FT3 and FT4 concentrations in any group of 10 diverse hypothyroid people, and substituting one LT4 or LT3 hormone pharmaceutical with another can have a wide range of results in blood (See L-T3 pharmaceutical equivalency, Part 2: New thyroid science).
Copyright fair dealing note
Quotation, paraphrase, and reproduction, annotation, and adaptation of graphs and tables from copyrighted scientific publications is acceptable within the terms of Canadian and US copyright “fair dealing” and “fair use” for purposes of education and review: See copyright law info
Gullo et al’s ratios and reference range
The health status of the study population
Click to expand section
In Gullo’s study, the health of the control group was described:
“Clinically euthyroid subjects with serum TSH<0.4 or >4.0 mU/L were excluded under suspicion of subclinical hyper- or hypothyroidism.
Subjects positive for anti-TPO and/or anti-Tg antibodies and/or with hyperechogenicity or pseudo-nodular pattern at thyroid ultrasound examination were also excluded.
As [the data set was subdivided] for athyreotic patients, also subjects in this group were subdivided by gender (F = 3,224, M = 651) and age (2,927<60 y and 948≥60 y).
None of these control subjects had ever been treated with thyroid hormones or antithyroid drugs.”
The average FT3:FT4 ratios
In Gullo’s Table 2, the TSH reference range was divided into 5 levels. At every level, the controls’ FT3:FT4 ratio was 0.31 to 0.33 pmol/L.

The FT3:FT4 ratio’s reference range
The FT3:FT4 ratio was given a reference range and population average in Gullo et al, 2011 in pmol/L, where the 3,875+ untreated healthy controls were contrasted with 1,811+ treated patients.

Gullo’s healthy controls’ mean was 0.32 (IQR 0.27–0.37) pmol/L in a reference range from 0.20 to 0.50 pmol/L, while the LT4-treated patients had a lower ratio represented by the skewed standard deviation curve.
Strich’s 2016: Effects of age on FT3:FT4 ratio
This study was conducted in Israel using data from 527,564 blood samples between January 2011 and September 2013, which were all tested on the same commercial immunoassays “Cobas kits used on modular analytics E-170 analyzer, Roche Diagnostics.”
They then excluded 422,012 samples that were excluded “due to lack of one parameter, mostly FT3.”
First, a caution: There’s a temptation to presume that a lower FT3:FT4 is “healthier” just because it is “normal” for older people. However, compare the decrease seen in the first table with the lack of a ratio decrease in the final table, which shows data only among those with normal body weight for their age and sex.
The health status of their study population
Click to expand details
Exclusion criteria were:
- No samples with positive titres of anti-TPO or anti-TG antibodies often found in autoimmune thyroid disease.
- No samples in patients treated with levothyroxine (L-T4) thyroid hormone, anti-thyroid medications (methimazole, propylthioracil / PTU), recombinant thyrotropin (pharmaceutical TSH injected before radioiodine ablation), all antiepileptic drugs, lithium, or glucocorticoids.
- No samples that had TSH above 7.5 or below 0.2 mIU/L. This removed those most likely to be ill or to have permanently altered thyroid physiology (“virtually all patients between the upper normal limits and this level have been shown to revert back to ‘normal’”). The expanded range permitted some TSH levels beyond the statistical reference range to reveal the influence of lower and higher TSH levels.
After all exclusions, the main data set included all three hormone levels for 27,940 persons, including 10,227 males and 17,713 females. Most of the samples were from younger people, and the full age range was from 1 year to 110 years with a mean of 24.07 ± 16.25 years old.
Table 1: Full data set by age
Notice that the decades of age are distributed unevenly. They are in 10-year age groups until age 40, but the age groups 40-60 and 60-80 each cover 20 years of age. Given their full data range cited above, the >80 category includes persons up to age 110.

Remarkable patterns
- The FT3 and FT4 were both higher in younger children. Human development apparently demands more thyroid hormone in circulation.
- The FT3 is the only perfectly linear data set, significantly decreasing with age.
- Within each decade, until 60 years of age, as TSH increased from Q1 to Q4, the FT3:FT4 ratio also increased.
- However, after 60 years of age, the increase in TSH did not always result in an increase in ratio.
Anomaly: Age 60-80 in TSH Q1 stands out as an abnormally high FT3:FT4 ratio amid lower ratios.
Table 2: Ratios analyzed
Another table provides further statistical insight into this data set, revealing the strongest trends across age groups and ratios:

Remarkable patterns:
- The FT3:FT4 ratio remains almost steady per decade on average between ages 20 and 80, except for a mild dip in the 40-60 age range category.
- The FT3:FT4 ratio is high in youth and low in age, mainly because of the influence of TSH on the ratio.
- The most perfect trend exists between TSH and the FT3:FT4 ratio, which expresses the way in which TSH enhances the FT3:FT4 ratio in blood more strongly in youth than in age.
Strich and team’s analysis noted that
“In the pediatric and young adults, until age 40, there was a positive and significant correlation between TSH and FT3/FT4 ratio (r = 0.08; P < 0.001), but in the older groups, this correlation decreased to nil as age increased (from 0.04 to −0.08)
This trend, i.e. the decreasing correlation with age was linear and significant (r = −0.94, P = 0.02)”
Partial patterns:
- The TSH-FT3 trend only applies to age 60.
Strich and team write about the positive linear correlation, excluding the point at which it turned into a negative correlation:
“Until 30 years of age, there was a significant positive linear correlation of TSH with FT3 (r = 0.14; P < 0.001), while in the above 30 groups, no positive correlation was noted.”
They omitted the observation of the negative trend from age 30-40 to 40-60.
- The heat map reveals no trend in the TSH-FT4 relationship across all the decades, only a fall in the 40-60 age category.
However, Strich and team make this odd observation:
“There was a negative correlation between FT4 and TSH (r = −0.02, P = 0.01) up to age 80.”
This reveals the research team’s desire to overemphasize a correlation between TSH and FT4 across the decades where the correlation is weak. Notice that the “r” value of -0.02 is very far from anything (the value r = 1 means a perfect positive correlation and the value r = -1 means a perfect negative correlation), and therefore giving the “p” significance level for this nonsignificant correlation is pointless.
As for the influence of gender, the researchers write:
“In general, both FT4 and FT3 are slightly lower among females for each TSH quartile (data available upon request).”
No tables are provided, but Gullo’s 2011 data set reveals that the “slightly lower” levels and ratio are a very slight difference across the sexes (for example, 0.31 versus 0.32). It is not worth one’s time to request the data.
Strich’s data set only with normal BMI
Next, Strich and fellow researchers focused the data set only on the subgroup with normal BMI (body mass index). They did this “because of reports that these parameters can affect the thyrotropin–thyroid axis,” citing several prior studies.
Indeed, obesity and metabolic syndrome can influence TSH-FT3-FT4 relationships significantly (Wolfenbuttel et al, 2017). In addition, unhealthy aging may involve muscle wasting and loss of bone mineral density along with other illnesses and lower levels of physical activity.

Remarkable patterns:
- In this “healthier” data set, there was no longer a decline in the FT3:FT4 ratio after the age of 20.
- The odd anomaly in the TSH Q1 of the age group 40-60 years of age was greatly minimized.
Strich’s discussion section provided insights into the normal HPT axis’s “effect of increasing TSH on FT3/FT4 ratio”:
“We have previously suggested that this phenomenon could be an in vivo reflection of the previously reported increase in the in vitro activity of deiodinases in response to increasing TSH concentrations.”
In other words, previous laboratory bench studies (in vitro) on tissues revealed that deiodinases D1 and D2 increase activity in response to increasing TSH concentrations, and this new data shows that the TSH effect on FT3:FT4 ratio exists in living organisms as well.
As for the influence of age, Strich and team reason that if rat studies represent the same phenomenon in humans, it is likely largely due to D1 decreasing with age, not receptor sensitivity decreasing with age:
“In aging rats, there are data showing a decline in the activity of type 1 deiodinase [D1] but no similar decline in thyroid hormone receptor expression or activity.”
A critique of Strich’s interpretations in the discussion section
This is a digression. Click to reveal if you are interested.
The research team makes puzzling and contradictory observations about the decrease in FT3:FT4 ratio in aging.
People from families with longevity, as well as older people in good health, often have a higher TSH and lower FT3 and FT4 levels. Therefore, Strich and team observe that
“If reduced metabolism is in fact a protective mechanism, it may explain why people who have lower FT4 and FT3 and also have a lower FT3/FT4 ratio are still alive at an older age.”
However, the data set with normal BMI shows no significant reduction in FT3:FT4 ratio after age 20. This removes the need for any hypothesis about reduced metabolism being “a protective mechanism.”
Why imply that a low FT3:FT4 ratio is protective?
Strich correctly note that in other studies of fetal tissues and damaged tissues deiodinase type 3 (D3) is upregulated.
However, they then gently imply that because this D3 deiodinase “protects fetal tissues,” it is also protecting damaged tissues and elderly people’s tissues:
“This deiodinase degrades T4 intracellularly to reverse T3, and one could speculate that increasing degradation of this type with age would dampen the effect of TSH on the FT3/TF4 ratio because more T4 is degraded to reverse T3 instead of to T3.”
There is no need to imply that TSH’s effect needs to be dampened in age, because
- Again, the FT3:FT4 ratio did not decrease in age in populations with normal BMI.
- Also, a higher FT3:FT4 ratio is not damaging in young people who have very high ratios.
- And, as mentioned above, healthy elderly persons do not have damaged tissues to protect with a lower ratio.
The direct opposite of the idea that a lower FT3:FT4 ratio is “protective” outside of fetal life is found in other research. Consider the association of low T3 syndrome (low FT3:FT4 ratio and high RT3) with high rates of morbidity and mortality in almost every type of chronic disease (Bianco et al, 2019; and see “Ataoglu: Low T3 in critical illness is deadly, and adding high T4 is worse.“).
Doctors must learn how to distinguish between acute and chronic low T3. Different conditions apply to the often inevitable and temporary low T3 state in the “acute” phase of nonthyroidal illness syndrome (NTIS) and the pathology of “chronic” low T3 in a state of nonthyroidal illness (Van den Berghe, 2014).
Reading the insights from Van den Berghe and others regarding NTIS recovery leads to a more rational conclusion: When the body is ready to recover, if either the TSH fails to rise, or a rising TSH fails to stimulate replenishment of FT3 from the thyroid (long before RT3 levels fall and metabolic imbalance is corrected), then the health outcomes are dismal. Essentially, the body’s failure to recover adequate FT3 and FT3:FT4 ratios when the body requires FT3 to heal its tissues is seemingly what prevents recovery of tissues.
Other research directly contradicts the implication that “damaged” tissues in diseases like cancer are protected by lower T3 levels binding to receptors in cells. Higher-normal levels of RT3 and FT4 can send signals on a very different receptor on the cell membrane, the integrin receptor, and there they can cause cancer to proliferate (See “Cancer scientists point finger at T4 & RT3 hormones.”)
Based on the deeply discerning level of cancer-thyroid research by Davis and team, one must learn to distinguish the genomic receptor signaling of FT3 in the nucleus, which is often benign, from FT4 and RT3 non-genomic signaling at the cell membrane, which can become pathological under certain states of illness.
Anderson’s ratio of average FT3 to average FT4
In Anderson’s 2020 article, the research team’s focus was on atrial fibrillation risk at various levels of FT4, TSH, and FT3. (See our review of their article: “Anderson, 2020: Thyroid hormones and atrial fibrillation.”
Not a true “average ratio”
This study did not calculate each person’s FT3:FT4 ratio and then average the ratios. Therefore, what you see below is a “ratio of averages,” rather than a true “average ratio.”
Nevertheless, perhaps because the results included so many individuals, the mathematical calculations of the “ratios of averages” appear to map on to Gullo’s and Strich’s data quite well.
The health status of the study population
Click to reveal section
The only exclusion was persons treated with levothyroxine (LT4).
This was not a healthy cohort, because they desired to discover associations with prevalence rates of cardiovascular disease and other chronic conditions itemized in their appendix.
However, their use of the reference range boundaries enabled the data set to exclude high and low values.
The normal quartile values are highlighted in the table below.
Ratios of average FT3 and FT4 across normal quartiles
Their raw unadjusted data set revealed the way in which using TSH levels as a lens usually results in flattening the FT3:FT4 ratio within each quartile (middle row).
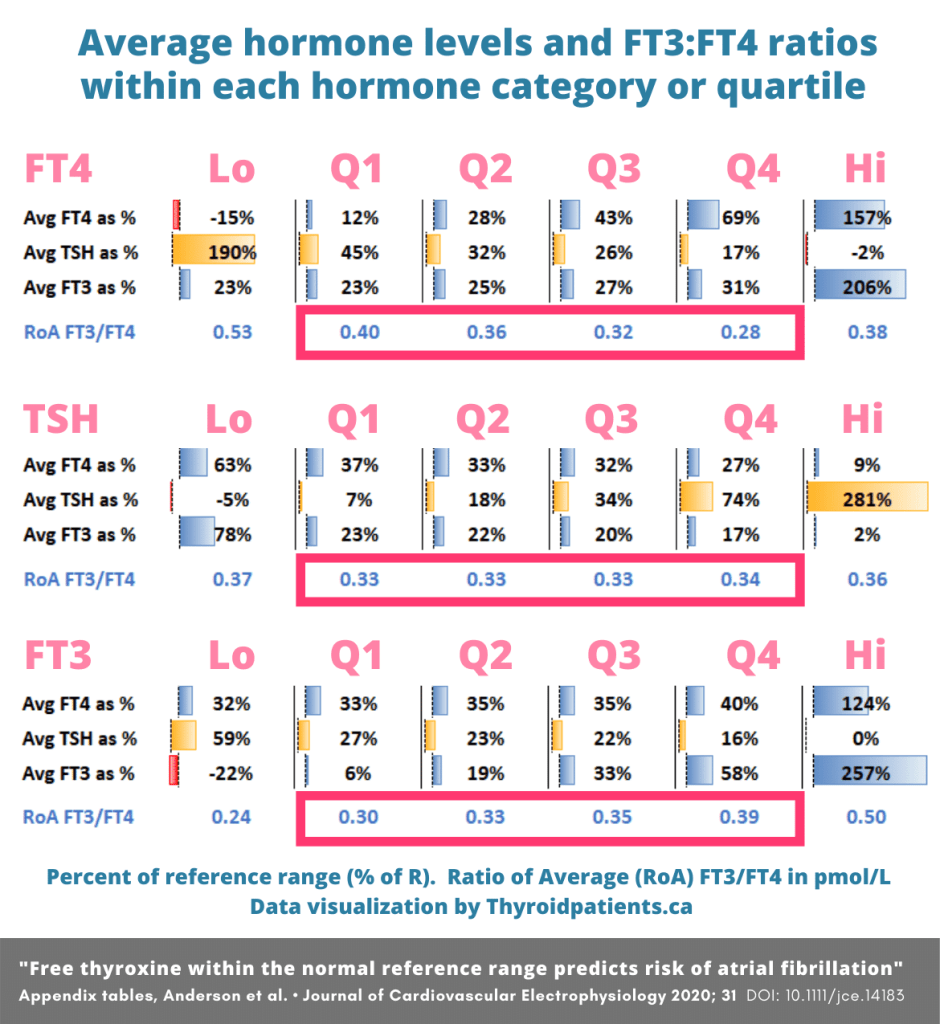
Ratios in the middle row, Normal TSH quartiles:
- TSH in Q1: Lowest TSH in reference, ratio normal
- TSH in Q4: Highest TSH in reference, ratio normal
In the middle row, the average TSH per quartile ranged widely, from 7% to 74% of reference (67% variation), since the range of TSH was the basis on which the quartiles were formed. However:
- FT4 varied little across the row, only decreasing from 37% to 27% from Q1 to Q4 (10% variation), hardly changing at all from Q2 to Q3.
- Likewise, the FT3 varied even less across the row, from 23% to 17% from Q1 to Q4 (only 6% variation), hardly changing at all between Q1 and Q3.
- Thirdly, the ratio of average FT3:FT4 held almost constant across the row, from 0.33 to 0.34. This is because the ratio of the average FT3 and average FT4 conceal all distinctions among individuals and the true range of ratios within each quartile.
Anderson’s ratios beyond the TSH normal quartiles
This data analysis includes people who have subclinical or overt thyroid disease:
Click to reveal if you are interested.
The low TSH and high TSH levels both had higher FT3:FT4 ratios. This is because the most common cause of hyperthyroidism, Graves’ disease, has the TSH-receptor stimulating antibody (TSAb). The TSH may be low or absent, but the TSH receptor is not empty. In fact, it is the opposite. The TSH receptor is bound to an antibody that can be more potent than TSH itself. The antibody elevates the FT3:FT4 ratio in blood and lowers TSH secretion rates. (See Research: TSAb antibody and TSH)
As you can see, when you view the data set through the lens of FT3 or FT4 quartiles, a wider range of FT3:FT4 ratios will appear.
The top row, FT4, has the widest range of FT3:FT4 ratios within the normal range. This is largely because of two principles in thyroid biology:
- Deiodinase type 2 (D2) is less efficient at converting T4 hormone to T3 when it is abundant in bloodstream, and is more efficient at conversion when T4 levels are lower, due to the vulnerability of D2 enzyme to “ubiquitination.” (See a review at “Ubiquitination: The glass ceiling of T4 monotherapy.”)
- TSH generally has an inverse relationship to FT4 but not to FT3. When FT4 is low, TSH rises to add TSH-Receptor stimulation to the thyroid gland, thereby enhancing the ratio of T3:T4 secreted by the thyroid gland, as explained above.
Again, the High TSH and High FT3 categories have high FT3:FT4 ratios because TSH-receptor signaling levels are high in Graves’ hyperthyroidism despite lower TSH concentrations of TSH, as explained above.
Summary: What is the normal FT3:FT4 range and average?
Gullo and team discovered that the average ratio among healthy controls throughout the normal TSH range (divided into quintiles) is approximately 0.32-0.33 per quintile in a population with an average age of 49 years.
- Anderson’s 2020 TSH data set coincidentally fits almost perfectly with Gullo’s range and average. Mid-range ratios of average FT3:FT4 in the table are approximately 0.33 mol/mol for all four normal TSH quartiles.
- This is a very narrow average. The average conceals the full range of individual diversity within any given quartile or quintile of TSH.
Gullo and team found the reference range for the ratio was from 0.20 to 0.50 pmol/L. This reference range is calculated, like most laboratory ranges, as the 95% interval, omitting the 2.5% high and 2.5% low outliers. It represents the wide range of normal human variation in the ratio.
- In Anderson et al’s 2020 data, including values outside the normal TSH reference range, the lowest ratio was 0.24 and highest ratio was 0.53. This is shifted only slightly higher than Gullo’s reference range of 0.20 to 0.50.
- Note that Anderson’s data set was not screened for overall health or thyroid health because the data set explored associations with disease, even within the reference range.
In addition, Strich found that the FT3:FT4 ratio decreased decade by decade from childhood to advanced age, but did not change as much between age 30 and 60. But when excluding all persons who had an abnormal weight for their age, the ratios narrowed down further, maintaining the average of 0.33 after age 20 even into the 80s.
- Anderson’s ratio average within TSH Normal Q1-Q4 is 0.3325 and the average age of the population was 64.9 years. This data fits within Strich et al’s average ratio within TSH Normal Q1-Q4: it was 0.345 in the 60-70-year-olds category.
- Gullo’s average ratios were 0.32 for all healthy controls, with an average age of 49 years. The control subjects’ sex and age data were F = 3,224, M= 651 and 2,927 <60 y and 948 >60 y.
- For women in Gullo’s study, the ratio was only slightly lower (0.32) than it was among men (0.33), which fits with Strich’s findings for sex differences.
- For persons over 60 in Gullo’s study, the ratio was slightly lower (0.30 for older women, 0.31 for older men). This fits with Strich’s findings of a mildly reduced ratio in older age groups when including all persons regardless of their body weight.
When measured in pmol/L, the ratio is consistent and comparable among three large population studies: Gullo et al, 2011’s healthy control group of 3,875 people, Strich et al, 2016’s study of 27,940 untreated patients at all ages, and Anderson et al, 2020’s appendix data set of 26, 524 patients who had all three tests, TSH, FT4 and FT3 completed.
Therefore, these FT3:FT4 ratio findings can assist in identifying metabolic dysfunctions and HPT axis interferences that may otherwise be misdiagnosed or overlooked.
In health, the average FT3:FT4 ratio and range at various levels of TSH is an indicator of the normal and healthy thyroid gland and thyroid hormone metabolism to TSH, and the TSH’s normal response to thyroid hormones.
- These hormone relationships are often abnormal in various types of thyroid disease and systemic illnesses. The TSH-FT3:FT4 relationship will also distort if the pituitary and hypothalamus’ co-adjustment of TSH secretion is abnormal.
In health, the presence of a normal FT3:FT4 ratio reflects the efficiency of the three deiodinases D1, D2, and D3 cooperation to metabolize thyroid hormones at various rates throughout all tissues in the body at various ages.
- The ratio may be abnormal if deiodinase genetic polymorphisms hinder, if substances or medications interfere, or if severe illness distorts the normal balance among the D1, D2 and D3 enzymes.
The ratio of FT3:FT4 can be judged low, normal, or high. Science provides us with a reference range for the ratio’s normal values at various levels of TSH, at various ages, with slight differences between males and females.
I also discuss the reliability and precision of testing FT3 and FT4 via common immunoassays available at laboratories today. This understanding is essential to assessing the validity and precision of the ratio calculation in everyday clinical practice.
I conclude by showing that the FT3:FT4 ratio has many potential practical uses: It can fine-tune diagnosis, reveal interferences, and in future, it may light the pathway to individually optimized therapeutic ratios in thyroid disease and other chronic diseases.
Metabolic principles taught by ratios
1. The FT3:FT4 ratio must always be anchored in a FT4 level.
In the TSH-driven thyroid hormone economy, T4 hormone will always be more abundant in blood than T3 hormone. The FT4 reference range is approximately 10-25 pmol/L while the FT3 reference range is approximately 3-6.5 pmol/L.
The FT4 level, by being converted to T3 at a variable rate, yields part of the T3 (FT3) level in blood. Most, but not all, of the untreated person’s circulating T3 (FT3) supply will be derived from intracellular T4-T3 conversion, as cells engage in 2-way exchange of thyroid hormones with blood. The body’s rate of T4-T3 conversion is always “topped up” by a flexible amount of T3 (FT3) from daily thyroidal secretion.
Therefore, at any given FT4 level, FT3 levels are “anchored” to their major metabolic donor, FT4. As the TSH-guided thyroid gland and peripheral metabolism work together, the body’s T4-T3 conversion rate plus thyroidal FT3 yield a total supply of T3 hormone to cellular receptors.
In addition, FT4 also performs some limited types of non-genomic signaling at the “integrin” cell membrane receptor on some cells in tumors, blood vessels, and immune system. As of 2019, scientists have also learned that the secondary metabolite of T4, Reverse T3 (RT3) is in fact an actively signaling hormone at this receptor. See our review at “Cancer scientists point finger at T4 & RT3 hormones.”
2. Low FT3:FT4 ratios and low FT3 are more pathological than an isolated low FT4.
This is because T3 hormone is the most potent signaling hormone in the nuclear receptor and in mitochondria.
This basic principle is most profoundly illustrated by T3 monotherapy, in which sufficient FT3 supply, often above the FT3 reference range, enables a hypothyroid individual to thrive on T3 alone, in the absence of FT4 from blood.
Higher FT4 cannot compensate for lower FT3, but a higher FT3 can compensate for lower FT4.
Are today’s FT3 and FT4 laboratory tests precise and reliable enough to yield this ratio?
Yes. Most manufacturers’ FT3 immunoassays are trustworthy for routine clinical and diagnostic purposes, especially when one understands the test’s technical performance when compared with FT4 assays.
Naive critics often fail to distinguish between technical precision and reliability on the one hand, and poor standardization among manufacturers and methods on the other hand.
The research on FT3:FT4 ratios above has been done using different standard immunoassays (Gullo used an unnamed assay in Italy; Strich used Roche; Anderson used Abbott) and despite their various technical shortcomings, they are remarkably in concordance with each other.
For advanced readers: Click to expand
According to an international committee that assessed standardization of thyroid hormone assays after the newer immunoassays were in circulation, both FT3 and FT4 assays are approximately equal in reliability and precision.
However, the assay manufacturers have not yet calibrated their absolute results to the gold standard LCMS method. (Thienpont et al, 2010). They usually vary less than 11% from the standard assay for calibration, and <11% is the target.
“Correlation coefficients to the cRMPs [conventional reference measurement procedure] ranged for FT4 (FT3) from 0.92 to 0.78 (0.88 to 0.30).
Within-run and total imprecision ranged for FT4 (FT3) from 1.0% to 11.1% (1.8% to 9.4%) and 1.5% to 14.1% (2.4% to 10.0%), respectively.
Approximately half of the manufacturers matched the internal QC [quality control] targets within ~5%”
(Thienpont et al, 2010)

The assays with lower means will normally have a lower reference range to suit each manufacturer’s technological bias. Assay-specific reference ranges are necessary to prevent inaccurate diagnosis.
Of course, the FT3 assay is more likely to be biased too low on the low end of reference when samples from unhealthy people (with nonthyroidal illness and persons on levothyroxine (LT4) monotherapy) are included in the calculation of reference ranges.
However, the use of anonymous or insufficiently screened samples is an error in the laboratory’s reference range research methodology, not a problem with the technical quality of the assay.
Another protection against assay bias is a clinician’s knowledge. A physician should be aware of the extremely tall and non-skewed curve of healthy FT3 values (almost 50% of healthy people are near mid-range) and FT3 values by age (see Strich et al’s data above). This knowledge can aid in interpreting low-normal FT3 values as unusual for a person’s age and concurrent FT4 level.

To assess the quality of FT3 and FT4 assays, one can also look at the methods sections of research articles that rely on these assays. Responsible researchers using FT3 and FT4 assays, such as those by Hoermann and team, will always report on the reliability and precision of their FT3 assays. They generally find them to be of good quality in comparison to FT4 and TSH assays.
Given the relatively equal quality of the two assays across many manufacturers, if one trusts FT4 to assist in diagnosis when TSH is in the subclinical zones, one should also trust the FT3 assay to refine diagnosis and to adjust therapy.
When measuring FT4, it makes sense to test Free T3 so that a metabolically meaningful ratio can be calculated.
Thienpont and team concluded, for both Free T4 and Free T3 assays,
“a majority of assays had acceptable quality of performance when measuring samples from nondiseased individuals;
however, some assays would benefit from improved precision, within-run stability, and between-run consistency.”
Therefore, despite the “acceptable quality,” improvements are still necessary.
In recent years, for example, it appears some immunoassays may have a pharmaceutical manufactuer’s bias, and/or poor performance and recalls, such as the Abbott Architect assay has in the past: See “Abbott Laboratories’ reference range normalizes lower Free T3” and “The Abbott Free T3 test kit recall of 2018-2019”
Are FT3, FT4 and TSH levels stable enough over time to assess this ratio?
Yes, especially if you know what time of day the TSH was tested. The TSH fluctuates far more widely than either FT3 or FT4 do during standard laboratory testing hours.
For advanced readers: Click to expand
Circadian rhythms of TSH and FT3
The wide circadian rhythms of TSH can interfere with accurate TSH-based diagnosis, but narrower FT3 rhythms enable FT3:FT4 ratio calculation.
See posts our that review circadian rhythm research:
- “Circadian rhythms of TSH, Free T4 and Free T3 in thyroid health”
- “The significance of the TSH-FT3 circadian rhythm“
- “Q&A. Dosing T3 in light of circadian rhythm“
As the nightly peak is reached in TSH levels, the FT3 echoes its rise, achieving the highest FT3 levels while we sleep. The rise in FT3 in the hours before rest and the high FT3 levels at night work in synergy with many other hormones that peak at these times in the 24-hour cycle. (People who dose thyroid hormones either lack a FT3 rhythm, or have a different rhythm based on dosing times.)
In the healthy HPT axis, FT3 drops in the morning hours, but the change during these hours covers a very small percentage of the FT3 reference range.
Naturally in people with healthy TSH-driven thyroids, the FT3:FT4 ratio is higher during the night than it is during the day. However, the ratio used in clinical practice and research is based on daytime measurements during laboratory hours.
Stability of FT3 over longer periods of time
Abdalla & Bianco (2014) point out the stability and precise adjustment of T3 levels over days, weeks and months in healthy people, a stability that is more remarkable than it is for TSH and T4:
Serum TT3 and FT3 exhibit minimal circadian rhythmicity that is due to a nocturnal increase in TSH secretion.
Otherwise, serum T3 is remarkably stable over periods of days, weeks or months in healthy adult individuals, despite a relatively short half-life (approximately 12–18 h).
The healthy human body optimizes its individualized FT3:FT4 ratio along with its levels of FT3 and FT4 within a very narrow band within the reference range.
One person’s “optimal” location in reference will be very different from another’s. (See data in the post “Individual thyroid ranges are far narrower than lab ranges“).

How does the body maintain or alter this ratio?
In the bloodstream, the ongoing rate of T4-T3 metabolism and transport will add FT3 to the supply from variable secretion rates and ratios from the thyroid gland, plus any pulsatile absorption from daily hormone dosing.
As TSH rises, the stimulation of a thyroid increases, and the levels of FT3 and FT4 as well as the FT3:FT4 ratio normally rise (T3 rises more than T4 does) in a state of gland health and metabolic health.
For advanced readers: Click to expand
Two things happen in healthy thyroid tissue as TSH rises:
- TSH-receptor stimulation preferentially stimulates T3 synthesis in the thyroid gland, adjusting the ratio of thyroidal secretion (Citterio et al, 2017; see our article “T3 is not always converted from T4: De novo T3 synthesis“.)
- TSH-receptor stimulation in the thyroid gland also stimulates D1 and D2 enzymes that convert T4 to T3 as blood flows through thyroid tissue.
When these two rates (synthesis + internal thyroid hormone conversion) are combined, it yields the “secretion ratio” of hormones released from the thyroid gland into circulation. By this means, TSH stimulation increases the T3:T4 ratio produced per unit of TSH, enhancing FT3 in blood. (Laurberg, 1984)
This T3 boosting function of TSH in the thyroid gland is called the “TSH-T3 shunt” (Berberich et al, 2018).
TSH-receptor signaling also simultaneously enhances the D1 and D2 enzymes found throughout the rest of the human body, since these enzymes are expressed to varying degrees in every organ and tissue.
The healthy thyroid gland is capable of compensating for shortfalls and excesses in thyroid hormone metabolism. (See tables and data in “Thyroid T3 secretion compensates for T4-T3 conversion“).
The widely variable rate and ratio of thyroidal secretion is what enables the human body to maintain and recover individually-customized, steady optimal FT3 levels and FT3:FT4 ratios in blood day to day, week to week, and month to month despite metabolic challenges, except for periods of severe illness.
The thyroid gland’s secretion flexibility within the TSH reference range is wider than many scientists have believed. The widespread, often un-cited mantra of the thyroid gland secreting only 20% of a person’s daily T3 supply, and of the thyroid gland secreting a 16:1 ratio of T4 to T3 in mcg/day (or 14:1 molar ratio), is based on a gross misinterpretation of the data in the main source, Pilo et al, 1990, that provided these average ratios in 14 subjects who had a TSH between 1 and 2 mU/L. (See a science-based critique of this idea of a single healthy ratio in our post “Meet a person with the perfect T3:T4 thyroid secretion ratio“)
More than one research study during the early era of intense “kinetic” studies of thyroid hormone economy show that thyroidal T3:T4 secretion ratios as well as rates vary widely among healthy individuals. (Laurberg et al, 1984)
This flexible ratio of secretion enables the thyroid gland to lead and support recovery from nonthyroidal illness syndrome (NTIS). During recovery, TSH can rise temporarily above reference range to replenish normal hormone supply before normal deiodinase function is re-established (Bacci et al, 1982; Brent et al 1986; Feelders et al, 1999).
How can the FT3:FT4 ratio enable the calculation of global T4-T3 conversion rate?
The bloodstream FT3:FT4 ratio reflects not only T4 and T3 supply but also the net product of different rates of T4-T3 conversion in tissues and organs throughout the body, including thyroid gland tissue.
The global rate of T4-T3 conversion is reflected in the FT3:FT4 ratio. No other biomarker yet discovered can come closer to assessing the status or bias of overall thyroid hormone metabolism.
For advanced readers: Click to expand
First, unbound (free) FT4 and FT3 are carried into cells on a wide variety of transporter proteins.
Next, T4-T3 conversion occurs inside cells that express D1 or D2 enzyme. In cells that express D1 or D3 enzyme, T4 can be converted to RT3 and T3 can be converted to inactive forms of T2. (D1 can perform both metabolic actions.)
After binding to receptors, or failing to bind to them, thyroid hormones are continually being transported out of cells at the same rate that hormones enter the cell, to keep equilibrium within the cell and surrounding tissues and blood (Bianco et al, 2019).
In this way, cells “donate” much of their metabolized thyroid hormones back to the bloodstream, along with any T3 and T4 that was not transformed by a deiodinase while flowing through the cell.
The 2-way exchange of hormone between bloodstream and cells happens every minute of every day, but some tissues may exchange hormone at a slower rate (bones, for example).
Each tissue in the body converts T4 and T3 at different rates to adapt bloodstream hormone levels to that tissue’s current metabolic needs. Some tissues are more efficient at converting FT4 than others that depend more on FT3 supply.
The FT3:FT4 ratio can be used to calculate a more refined result than the ratio itself, called “Global Deiodinase efficiency” (GD), using the free SPINA-Thyr endocrinology research application (Midgley et al, 2015; Dietrich et al, 2016; See an overview of the program: “Analyze thyroid lab results using SPINA-Thyr.”)
The “GD” quotient is based on the understanding that there is a continual two-way exchange of circulating thyroid hormones with cells that metabolize thyroid hormones throughout the human body, and a clearance rate and hormone binding rate that corresponds to the level of thyroid hormones in blood.
Differential diagnoses enabled by the FT3:FT4 ratio in light of TSH
When healthy thyroid tissue expressing D1 and D2 is present, a higher TSH usually enhances the T3 side of the FT3:FT4 ratio.
Before treatment with thyroid hormone, when a normal or high TSH does not result in the normal or elevated FT3:FT4 ratio expected, something is amiss with
- thyroid gland function,
- poor D1 or D2 deiodinase function or elevated D3 deiodinase, and/or
- poor TSH hormone quality or TSHR signaling interference.
As for TSHR signaling interference, TSH concentrations in blood do not always correspond with the rates of TSH-receptor (TSHR) signaling because substances other than TSH, like TSH-receptor antibodies, can bind to the TSHR and boost or invert its signal.
Not just TSH concentration, but TSH-receptor signaling, influences the FT3:FT4 ratio.
When the FT3:FT4 ratio is inappropriate to TSH concentrations, the mismatch enables further diagnostic discernment of abnormalities in thyroid hormone supply and the TSH secretion’s response to that supply.
For example,
Autoimmune hyperthyroidism, or…?
An inappropriately normal or low FT3:FT4 ratio in the presence of low or suppressed TSH can distinguish special forms of hyperthyroidism that do not elevate the ratio from the more common forms that do.
- Excess human chorionic gonadotropin (hCG) hormone stimulation of TSH receptors in pregnancy, often raises FT4 and FT3 together, or elevates mainly Free T4 (See “Pregnancy thyrotoxicosis vs just a low TSH due to hCG hormone).
- Graves’ TSH receptor-stimulating antibodies have the unique ability to enhance D1 deiodinase (most dominant in thyroid, liver, and kidney) (Chen et al, 2018) via enhanced TSHR signaling, even when TSH is absent from circulation. Patients with T3-dominant ratios are more likely to have not only higher titres of TSAb but also lower or absent concurrent titres of blocking antibody TBAb (McLachlan & Rapoport, 2013).
- Autonomous functioning thyroid nodules can elevate the ratio, as well (Wong & Volpe, 1981; Ridgway et al, 1973), even when TSAb antibody is not present.
“Subclinical” hypothyroidism, or…?
An inappropriately normal FT3:FT4 ratio in the presence of mildly elevated TSH can distinguish early thyroid gland failure from central hypothyroidism or less bioactive forms of TSH. These other forms of hypothyroidism may require further investigation to prevent other health problems.
- During early thyroid failure in Hashimoto’s thyroiditis, elevated TSH secretion will raise the FT3:FT4 ratio significantly via T3 synthesis and upregulation of D1 and D2 in remaining functional thyroid tissue (Hoermann et al, 2020). If the FT3 is enough to compensate for the lower FT4, the patient may remain without hypothyroid symptoms until the gland is further damaged.
- In central hypothyroidism of the tertiary (hypothalamc) variety, the quality of TSH is compromised by low hypothalamic TRH secretion or the inability of the pituitary to receive TRH signals (Persani et al, 2019). See “Why is central hypothyroidism so difficult to diagnose?” Such patients require investigation of other hypothalamic and pituitary hormone deficiencies that can cause health problems beyond hypothyroidism.
- Macro-TSH, anti-mouse antibodies and IgG-associated TSH have been found in cases of TSH over 2.5 mU/L among infertile women. (Hattori et al, 2018). These cases may have been infertile due to insufficient thyroid gland stimulation and lowered hormone secretion.
Hashimoto’s thyroiditis or …?
An abnormally low or normal FT3:FT4 ratio in the presence of extremely high TSH > 80 mU/L can distinguish Atrophic thyroiditis and/or Blocking hypothyroidism (caused by the TSH-receptor blocking antibody, TBAb) from pure Hashimoto’s thyroiditis without TBAb titres.
In “Blocking hypothyroidism” (Tagami et al, 2019), high levels of TSH may be unable to stimulate even a fully healthy thyroid gland (revealed by ultrasound) to secrete enough T4 and T3 hormones, resulting in an abnormally non-elevated FT3:FT4 ratio.
Diagnosis affects treatment because some cases of blocking hypothyroidism may lead to full remission shortly after treatment, and antibodies may later return or fluctuate between stimulating (Graves’ hyperthyroidism) and blocking hypothyroidism , potentially causing unstable thyroid therapy (Takasu et al, 2012). This potent, volatile form of thyroid autoimmunity remains largely undiagnosed in a significant population of “Hashimoto’s” patients (For numeric estimates, see “Overlooked: How many Hashimoto’s patients with TSH-Receptor antibodies?“).
FT3:FT4 ratio is lowered in TBAb antibody activity because TSH receptor stimulation is not just blocked but lowered below baseline TSHR signal levels, downregulating D2 and D1 enzymes in the pituitary, in the thyroid, and throughout the body. The TBAb antibody is a TSH-receptor “inverse agonist” not just an inert receptor blocker (McLachlan & Rapoport, 2013).
The FT3:FT4 ratio supports antibody detection because many newer TRAb antibody tests are methodologically unable to report the titre of TBAb blocking antibodies when they are present alongside or instead of TSAb antibodies (McLachlan & Rapoport, 2013; Lytton et al, 2018).
Ultrasound thyroid measurements, lack of goiter, and unreasonably fluctuating TSH-FT4 relationships also confirm diagnosis. The antibody can even prevent goiter (thyroid swelling) normally seen when TSH is elevated, but cases with goiter are more likely to lead to remission (Takasu et al, 2012).
For more information, see “The Spectrum of Thyroid Autoimmunity” and “The THIRD type of autoimmune thyroid disease: Atrophic Thyroiditis“
Avoid misapplications of the FT3:FT4 ratio
Learning about FT3:FT4 ratios requires more than understanding and imitating population norms and averages. The science of the normal ratio is just a starting place.
The key is in the title of this post: “Large populations” have a pattern that enables diagnosis.
The normal pattern and range exists because of normal thyroid physiology and normal cellular signaling pathways.
Diagnosis is about understanding which aspects of the normal system have failed. However, diagnosis does not tell us the way back to health.
Description of a healthy population is not a prescription for a healthy individual.
Even though average FT3:FT4 ratios exist in large, healthy populations when viewed through the lens of TSH quartiles, optimal thyroid hormone levels, if not FT3:FT4 ratios, are diverse for each thyroid-healthy individual in the untreated population (see “Individual thyroid ranges are far narrower than lab ranges“)
In addition to an optimal ratio, we each have an optimal level of T3 receptor signaling, a metabolic setpoint, even in health (Abdalla & Bianco, 2014).
The individually unique level in health is hidden behind a statistical norm.
FT3:FT4 ratios are individually optimized for each individual in health.
Biochemical norms and ranges of health aid interpretation and diagnosis.
After treatment, ranges should never be misused as treatment targets or treatment boundaries. Enforcing biochemical conformity on thyroid-disabled, treated people goes down the dangerous and unethical path of “biochemical bigotry.”
References
Click to reveal reference list
Abdalla, S. M., & Bianco, A. C. (2014). Defending plasma T3 is a biological priority. Clinical Endocrinology, 81(5), 633–641. https://doi.org/10.1111/cen.12538
Anderson, J. L., Jacobs, V., May, H. T., Bair, T. L., Benowitz, B. A., Lappe, D. L., Muhlestein, J. B., Knowlton, K. U., & Bunch, T. J. (2020). Free thyroxine within the normal reference range predicts risk of atrial fibrillation. Journal of Cardiovascular Electrophysiology, 31(1), 18–29. https://doi.org/10.1111/jce.14183
Bacci, V., Schussler, G. C., & Kaplan, T. B. (1982). The relationship between serum triiodothyronine and thyrotropin during systemic illness. The Journal of Clinical Endocrinology and Metabolism, 54(6), 1229–1235. https://doi.org/10.1210/jcem-54-6-1229
Bianco, A. C., Dumitrescu, A., Gereben, B., Ribeiro, M. O., Fonseca, T. L., Fernandes, G. W., & Bocco, B. M. L. C. (2019). Paradigms of Dynamic Control of Thyroid Hormone Signaling. Endocrine Reviews, 40(4), 1000–1047. https://doi.org/10.1210/er.2018-00275
Berberich, J., Dietrich, J. W., Hoermann, R., & Müller, M. A. (2018). Mathematical Modeling of the Pituitary–Thyroid Feedback Loop: Role of a TSH-T3-Shunt and Sensitivity Analysis. Frontiers in Endocrinology, 9. https://doi.org/10.3389/fendo.2018.00091
Brent, G. A., Hershman, J. M., & Braunstein, G. D. (1986). Patients with severe nonthyroidal illness and serum thyrotropin concentrations in the hypothyroid range. The American Journal of Medicine, 81(3), 463–466. https://doi.org/10.1016/0002-9343(86)90300-1
Chen, X., Zhou, Y., Zhou, M., Yin, Q., & Wang, S. (2018). Diagnostic Values of Free Triiodothyronine and Free Thyroxine and the Ratio of Free Triiodothyronine to Free Thyroxine in Thyrotoxicosis. International Journal of Endocrinology, 2018, 4836736. https://doi.org/10.1155/2018/4836736
Citterio, C. E., Veluswamy, B., Morgan, S. J., Galton, V. A., Banga, J. P., Atkins, S., Morishita, Y., Neumann, S., Latif, R., Gershengorn, M. C., Smith, T. J., & Arvan, P. (2017). De novo triiodothyronine formation from thyrocytes activated by thyroid-stimulating hormone. The Journal of Biological Chemistry, 292(37), 15434–15444. https://doi.org/10.1074/jbc.M117.784447
Davies, T. F., & Latif, R. (2019). Editorial: TSH Receptor and Autoimmunity. Frontiers in Endocrinology, 10, 19. https://doi.org/10.3389/fendo.2019.00019
Feelders, R. A., Swaak, A. J. G., Romijn, J. A., Eggermont, A. M. M., Tielens, E. T., Vreugdenhill, G., Endert, E., Eijk, H. G. van, & Berghout, A. (1999). Characteristics of recovery from the euthyroid sick syndrome induced by tumor necrosis factor alpha in cancer patients. Metabolism – Clinical and Experimental, 48(3), 324–329. https://doi.org/10.1016/S0026-0495(99)90080-X
Gullo, D., Latina, A., Frasca, F., Le Moli, R., Pellegriti, G., & Vigneri, R. (2011). Levothyroxine Monotherapy Cannot Guarantee Euthyroidism in All Athyreotic Patients. PLoS ONE, 6(8). https://doi.org/10.1371/journal.pone.0022552
Hattori, N., Aisaka, K., Chihara, K., & Shimatsu, A. (2018). Current Thyrotropin Immunoassays Recognize Macro-Thyrotropin Leading to Hyperthyrotropinemia in Females of Reproductive Age. Thyroid: Official Journal of the American Thyroid Association, 28(10), 1252–1260. https://doi.org/10.1089/thy.2017.0624
Hoermann, R., Pekker, M. J., Midgley, J. E. M., Larisch, R., & Dietrich, J. W. (2020). Triiodothyronine secretion in early thyroid failure: The adaptive response of central feedforward control. European Journal of Clinical Investigation, 50(2), e13192. https://doi.org/10.1111/eci.13192
Laurberg, P. (1984). Mechanisms governing the relative proportions of thyroxine and 3,5,3’-triiodothyronine in thyroid secretion. Metabolism: Clinical and Experimental, 33(4), 379–392. https://www.metabolismjournal.com/article/0026-0495(84)90203-8/fulltext
Lytton, S. D., Schluter, A., & Banga, P. J. (2018). Functional diagnostics for thyrotropin hormone receptor autoantibodies: Bioassays prevail over binding assays. Frontiers in Bioscience (Landmark Edition), 23, 2028–2043.
McLachlan, S. M., & Rapoport, B. (2013). Thyrotropin-Blocking Autoantibodies and Thyroid-Stimulating Autoantibodies: Potential Mechanisms Involved in the Pendulum Swinging from Hypothyroidism to Hyperthyroidism or Vice Versa. Thyroid, 23(1), 14–24. https://doi.org/10.1089/thy.2012.0374
Midgley, J. E. M., Larisch, R., Dietrich, J. W., & Hoermann, R. (2015). Variation in the biochemical response to l-thyroxine therapy and relationship with peripheral thyroid hormone conversion efficiency. Endocrine Connections, 4(4), 196–205. https://doi.org/10.1530/EC-15-0056
Persani, L., Cangiano, B., & Bonomi, M. (2019). The diagnosis and management of central hypothyroidism in 2018. Endocrine Connections. https://doi.org/10.1530/EC-18-0515
Ridgway, E. C., Weintraub, B. D., Cevallos, J. L., Rack, M. C., & Maloof, F. (1973). Suppression of Pituitary TSH Secretion in the Patient with a Hyperfunctioning Thyroid Nodule. Journal of Clinical Investigation, 52(11), 2783–2792. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC302546/
Silva, J. E., Dick, T. E., & Larsen, P. R. (1978). The contribution of local tissue thyroxine monodeiodination to the nuclear 3,5,3’-triiodothyronine in pituitary, liver, and kidney of euthyroid rats. Endocrinology, 103(4), 1196–1207. https://doi.org/10.1210/endo-103-4-1196
Strich, D., Karavani, G., Edri, S., & Gillis, D. (2016). TSH enhancement of FT4 to FT3 conversion is age dependent. European Journal of Endocrinology, 175(1), 49–54. https://doi.org/10.1530/EJE-16-0007
Tagami, T., Hiroshima-Hamanaka, K., Umakoshi, H., Tsuiki-Naruse, M., Kusakabe, T., Satoh-Asahara, N., Shimatsu, A., & Moriyama, K. (2019). Experimental Reproduction of Dynamic Fluctuation of TSH Receptor-Binding Antibodies Between Stimulation and Inhibition. Journal of the Endocrine Society, 3(12), 2361–2373. https://doi.org/10.1210/js.2019-00012
Takasu, N., & Matsushita, M. (2012). Changes of TSH-Stimulation Blocking Antibody (TSBAb) and Thyroid Stimulating Antibody (TSAb) Over 10 Years in 34 TSBAb-Positive Patients with Hypothyroidism and in 98 TSAb-Positive Graves’ Patients with Hyperthyroidism: Reevaluation of TSBAb and TSAb in TSH-Receptor-Antibody (TRAb)-Positive Patients. Journal of Thyroid Research, 2012, 182176. https://doi.org/10.1155/2012/182176
Thienpont, L. M., Van Uytfanghe, K., Beastall, G., Faix, J. D., Ieiri, T., Miller, W. G., Nelson, J. C., Ronin, C., Ross, H. A., Thijssen, J. H., Toussaint, B., & IFCC Working Group on Standardization of Thyroid Function Tests. (2010). Report of the IFCC Working Group for Standardization of Thyroid Function Tests; part 2: Free thyroxine and free triiodothyronine. Clinical Chemistry, 56(6), 912–920. https://doi.org/10.1373/clinchem.2009.140194
Van den Berghe, G. (2014). Non-thyroidal illness in the ICU: A syndrome with different faces. Thyroid: Official Journal of the American Thyroid Association, 24(10), 1456–1465. https://doi.org/10.1089/thy.2014.0201
Wolffenbuttel, B. H. R., Wouters, H. J. C. M., Slagter, S. N., van Waateringe, R. P., Muller Kobold, A. C., van Vliet-Ostaptchouk, J. V., Links, T. P., & van der Klauw, M. M. (2017). Thyroid function and metabolic syndrome in the population-based LifeLines cohort study. BMC Endocrine Disorders, 17. https://doi.org/10.1186/s12902-017-0215-1
Wong, M. M., & Volpé, R. (1981). What is the best test for monitoring levothyroxine therapy? Canadian Medical Association Journal, 124(9), 1181–1183. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1705308/

Leave a public reply here, on our website.