
In health, what happens when your tissues don’t convert enough T4 into T3 hormone? Your healthy thyroid secretes more T3.
The healthy thyroid gland’s synthesis of T3 de novo (from raw materials of iodine and tyrosine) under the stimulation of a healthy TSH can maintain your circulating T3 supply. This prevent tissues from becoming hypothyroid when they can’t convert enough T4 into T3 locally.
This principle has been demonstrated in human models in Pilo’s study in 1990, and several other scientific studies. It is
“the capacity of the hypothalamic-pituitary-thyroid axis to compensate for the lack of peripheral T4-to-T3 conversion.”
(Marsili et al, 2011)
The healthy TSH-stimulated thyroid has the flexibility to secrete more T3, when needed, to compensate for poor peripheral T4-T3 conversion (in tissues beyond the thyroid).
Scientists who observed the mouse’s thyroid gland secretion adapt to life without D1 and D2 enzymes, who could not convert T4 to T3, emphasized this lesson:
“the importance of the thyroid per se as a source of T3.”
(Galton et al, 2009)
However, a common generalization expressed over and over in scientific articles dismisses the thyroid’s importance as a source of T3 in humans.
We are given the impression that peripheral T4-T3 conversion don’t vary much across individuals, and a human thyroid is not capable of adaptive compensation in its T3 secretion rate, because a reduction in conversion rate is unthinkable. The ratio for humans is expressed as if it is fixed in stone:
“20% of daily T3 comes from the thyroid,
80% from peripheral conversion of T4.”
This idea of an inflexible ratio is an inaccurate representation of statistical averages from a famous scientific research article from 1990. It’s a narrow-minded concept that requires correction from the article itself and from confirming studies.
This idea of limited thyroidal T3 secretion, of a fixed, inflexible thyroidal T3 secretion, is found all over thyroid therapy literature, such as in this passage in the American Thyroid Association (ATA) 2014 guidelines by Jonklaas et al:
“Approximately 85 mcg of T4 is secreted by the thyroid gland daily. Of the total daily T3 production of about 33 mcg in normal man, approximately 80% (about 26 mcg) arises from peripheral conversion from T4, and only about 20% (approximately 6.5 mcg) derives from direct thyroidal secretion (34).”
This passage begins by emphasizing T4 and its approximate contribution to daily T3 production rate, and ends by diminishing “only about 20%” from “thyroidal secretion.”
What is item “(34)” cited at the end of the passage? It is Alessandro Pilo and team’s famous 1990 kinetic study of T4 and T3 hormone economy in healthy humans (or as the ATA say, “normal man”).
Most people reading this passage will believe the ATA is providing accurate figures (85 mcg, 33 mcg, 26 mcg, and 6.5 mcg). No. They didn’t count on having a humble thyroid patient fact-checking their calculations.
Pilo’s study, which they cite, did NOT discover, as the ATA states, that “85 mcg of T4 is secreted by the thyroid gland daily.” Nor did it find that 20% was a good estimate of the rate of T3 secretion from all human thyroids. The ATA’s word “approximately” is no excuse. They give extremely precise numbers, and yet they are off by 15 micrograms of T4 based on Pilo’s article.
Why does it matter that they are off by 15 micrograms here or there?
Because such narrow calculations have become fundamental premises on which people have built many narrow, lopsided theories to defend their preferred therapy practices.
Thyroid therapy has become a game of numbers. When medicine tries to replace a flexible gland with a static dose of hormone, people’s health and well being are based on precise microgram doses, lab reference ranges, and theoretical concepts like ratios.
Our systems have to respect the wide ranges of individual variation that nature reveals and science has documented. If they misrepresent the wide variations in secretion and conversion rates as if they are narrow averages that apply to everyone, it can have devastating implications for individuals who aren’t statistically average.
How would you like to live with a suboptimal FT3 level for the rest of your life because your gullible doctor reveres the ATA and believes you are a “normal man” or woman who can live happily on a ratio of 85 mcg / day of LT4 to 6.5 mcg of LT3 as long as it normalizes your TSH?
What if, under the cover of the statistically normal TSH, this dosing strategy is not providing the T3/T4 ratio your thyroid gland would have provided in health?
The details from Pilo’s 1990 study are more accommodating of individual diversity than the ATA wants them to be.
In this old study of 14 healthy human beings, they estimated one person had 42% of their total T3 supply from their thyroid gland and 58% came from peripheral T4-T3 conversion.
- That’s not a 20/80 T3 secretion / conversion ratio. That’s more than 2x the 20% people are told.
In another patient in the same study, 6.5% came from their thyroid gland and 93.5% from peripheral conversion.
- That’s not a 20/80 ratio, either. That’s less than 1/2 the 20%.
Yet the idea of 20% of T3 supply from the thyroid is cited today over and over as if it is as universal as the law of gravity.
Sadly, squeezing a such a wide range of human variation into a static ratio is harmful when applied to millions of diverse individuals on thyroid therapy. It has caused great injury to those whose peripheral thyroid hormone metabolism produces a significant T3 deficit, people like the man whose thyroid had to step up its game and secrete 42% of his T3 supply. Not every woman or man fits the average “normal man” parameters.
Pilo’s study is by no means perfect, but its published data set is worthy of a detailed look in the new color-coded tables I provide in today’s post.
Summary / Abstract

Click here to expand and read. This summary was inserted after it was posted as a separate blog post.
Overview
Pilo’s 1990 study attempted to quantify the amount of T4 and T3 hormone secreted by the thyroid compared to how much T4 was metabolized to T3 throughout the body.
The study had key flaws in methodology and was limited by an incomplete, earlier understanding of the mechanisms of thyroid hormone transport and metabolism. Despite these flaws and limitations of their study, the detailed data and estimates published in Pilo’s article are still enlightening today.
In Pilo’s data tables, they break down the data for each of their 14 human subjects, 9 men and 5 women.
These data, when analyzed by re-sorting by columns in the data set, reveal ten important principles and applications:
#1. The average T3 from secretion, 20% and average T3 from peripheral conversion, 80%, do not represent the full range seen among the 14 people. The data ranged from 6.5% T3 from the thyroid gland to 42% from the thyroid gland. The statistical average of 20% was close to only one of the 14 people in the study.
#2. In the person who secreted 6.5% of their T3 from their thyroid gland, 93.5% of their supply had to come from peripheral conversion. Clearly and obviously, in healthy people, conversion rate compensates for lack of secretion rate. Each individual is unique in the population.
#3. If you remove the thyroid gland from the person who was secreting 42% of their daily T3 supply from their thyroid gland, in theory, you will steal 42% of their T3 from their body. Now their peripheral metabolism will supply 100%, but the 100% will represent a lower number of micrograms per day. Can their metabolism make up for the loss? Will the total amount of T3 in bloodstream be lower? How much lower? It all depends on the health of a person’s thyroid hormone metabolism: how well the individual can convert T4 to T3 without a thyroid gland.
#4. The data show that people who have the least quantity of T3 in circulation are the ones who secrete the most from their thyroid gland, and vice versa. However, the strong directly inverse relationship seen at the extremely low and extremely high end of T3 supply does not apply to the 7 people in the middle of the data set. Once again, the generalizations about the data as a whole cannot apply to the individual within the population.
#5. The anomalies in Pilo’s data are explained by thyroid homeostatic flux. Our thyroid hormone economy must temporarily invert the standard relationship between secretion and peripheral conversion to raise or lower the total T3 supply in blood. We need only look to our current scientific understanding of the healthy HPT axis (hypothalamus-pituitary-thyroid axis).
The thyroid hormone economy in an individual is capable of significant flux as the pituitary secretion of TSH raises and lowers our metabolic rate on demand. As the body adjusts its T3 blood supply higher in response to demand, the relationship between secreted T3 and peripherally converted T3 will invert in relationship to the total supply — more TSH will lead to more T3 secretion from the gland and relatively less peripheral conversion, and the total T3 in circulation will rise.
But after the new homeostasis has been achieved, the the TSH-T3 state will normalize, and those with the largest T3 supply in blood will once again reduce their T3 secretion from the thyroid gland. Negative feedback from T3 will lower the TSH.
The “stasis” pattern of “the more T3 in blood, the less secreted from the thyroid” will prevail in the population only because a larger number of people will be in a comfortable position of “homeostasis achieved” than the number of people in the process of a “metabolic transition.” During an upward or downward transition, the thyroidal secretion rate will be abnormally high or low in relation to the total supply of T3 in blood.
#6. If you remove the thyroid gland’s ability to raise its T3 secretion rate to raise T3 supply, you remove the engine that provides them with the metabolic flexibility they need to meet changing demands. They can neither increase their T3 supply nor lower their T3 supply by means of TSH-driven T3 secretion from the thyroid gland.
6A) Flexibility to meet demands of seasonal change. New research on thyroid metabolic flexibility in seasonal change has shown that the thyroidless population maintained on LT4 hormone dosing alone cannot meet the demand for more T3 needed in the colder winter season. While the healthy population gains T3 in winter, the thyroidless lose a significant amount of T3 in winter if thyroid hormone LT4 dose remains unchanged. They cannot keep up with the body’s demand for more T3 in winter because they cannot raise the T3 secretion rate from their thyroid, which is missing.
6B) Flexibility to meet demands during nonthyroidal illness. The risk to thyroid-disabled patients is apparent in the research on mortality risk during “Nonthyroidal illness syndrome” (NTIS), also called Low T3 syndrome. If you remove the thyroid gland, you remove the T3-secretion flexibility needed to recover from the T3 hormone deficit induced by NTIS.
In all human beings, regardless of their thyroid hormone health, a severe critical illness such as a heart attack can result in swift loss of T3 from blood as the body requires a swift reduction in metabolic rate. During this temporary low T3 phase, the body is protected from undue energy expenditure. It’s like a temporary state of hibernation.
However, at the point of recovery from NTIS, the point of the individual’s greatest T3 depletion, the TSH must rise to stimulate T3 repletion in proportion with a recovering T4-T3 conversion rate. Recovery of T3 enables organs and tissues to heal. Data show that patients with healthy thyroids whose T3 levels fall too low, who are unable to raise their TSH and replenish T3 supply, have a very high mortality rate. Among those who recover, the TSH-stimulated increase in the rate of T3 secretion from the thyroid gland is the main engine that revives and restores the T3 supply in blood and reestablishes the healthy thyroid hormone economy.
In theory, thyroidless people (and those with inability to secrete enough TSH) lack this engine to recover from NTIS. But our vulnerable population has been excluded from almost all studies of NTIS. Like Pilo’s study, researchers have only studied the healthy. They have only seen the NTIS metabolic flux and mortality risk in the thyroid-healthy population. They haven’t reasoned about or examined the long-term health risk in the thyroid-disabled population.
#8. Pilo’s data on TSH levels in relationship to T3 secretion and conversion rates shows no pattern or relationship at all. This lack of TSH-T3 relationship is metabolically significant. It reveals the driver of the metabolic flux we see in people who enjoy thyroid gland health. TSH is the driver of the shift from “metabolic transition” to the achievement of a new “metabolic homeostasis.”
While TSH is in the process of rising or TSH is in the process of falling, we will see exceptions to the classic principle of the negative feedback loop. TSH supply will temporarily invert its relationship to T3 secretion vs. T3 conversion.
Instead of trying to understand metabolic flux and disorder revealed in the data set between TSH and T3, scientists decided to ignore the messy data.
Scientists decided to theorize an incomplete, simplified HPT axis model that omitted the most powerful and essential T3 hormone. They chose to focus on the steady, standard, inverted relationship between TSH concentrations and T4 concentrations in blood.
Scientists also decided to simplify the model by emphasizing the statistical population reference range boundaries for TSH, T3 / Free T3, and T4 / Free T4. This preference to judge euthyroidism by reference range emphasizes the statistical norms of the population and conveniently obscures the extreme diversity of individuals.
Research has shown that the Free T3 concentration has the narrowest range of movement up and down within the reference range over time, and Free T3 concentrations are the most diverse from one individual to another. The messiness of TSH flux and T3 secretion/conversion compensation flux occur “behind the scenes.” Their flux is necessary to optimize T3 levels in blood, since T3 is the most powerful thyroid hormone.
#9. Pilo’s data gives us insight into the influence of sex and age on T3 secretion and T3 conversion rate averages. When his data is sorted by sex and age, the five females generally had the least T3 secretion from the thyroid, with the youngest secreting the most and the oldest secreting the least. Among the nine males, T3 secretion rates were much higher on average, but the data was very inconsistent by age.
Pilo’s averages for the population as a whole are weighted toward the nine men. With the exception of one man and one woman, the men had twice the T3 thyroidal secretion rate as the women.
The bias in Pilo’s data set makes it incorrect to generalize his statistical averages to the health of the thyroid-disabled population on thyroid therapy, most of whom are females over forty years of age.
It is time to re-do this study including both men and women at all ages.
We must also study a large population of people pre- and post-thyroidectomy to understand how T3 peripheral conversion adapts to the thyroidless state in all sexes and ages.
#10. Finally, we must question the degree to which Pilo’s research subjects had a healthy HPT axis and were truly healthy during the study. The research article did not rule out crucial features like mild central hypothyroidism, an undiagnosed chronic nonthyroidal illness, iodine excess or deficiency at the time baseline data was collected; nor the presence of supplements, medications and dietary factors that influence T4-T3 conversion rate.
Pilo’s team performed research before the genetic revolution and could not test for polymorphisms in DIO1 and DIO2 that can significantly affect T3 levels and secretion/conversion ratios.
The study did not measure any biomarkers of T3 signalling throughout the body such as heart rate, cholesterol, ankle reflex, or bone metabolism, so it was unable to determine whether a person’s T3 supply from T3 secretion or or T3 conversion was sufficient for their body’s needs.
#11. Solutions:
A. Respect T3 flexibility and diversity.
B. Change the mantra. No longer claim that the thyroid secretes 20% of our T3 supply and the peripheral metabolism converts 80% of our daily T3 supply. Change it to “A thyroid may secrete between 6% and 42% of total daily T3 supply to compensate for peripheral T4-T3 conversion rates.”
C. Study how to optimize each thyroid patient’s FT3 and FT4. Pilo’s study says nothing about how to maintain health in a thyroid hormone economy that is no longer driven by the partnership between TSH and a flexibly secreting thyroid gland. Yet his study is being misused to limit and oversimplify thyroid therapy today.
Pilo’s data prove that a programme of mere “TSH normalization” in thyroid therapy cannot flexibly adjust T3 levels in patients to meet their diverse individual needs or their changing metabolic demands. Listen to the research already published on this topic. Continue to perform more research on thyroid patients’ adaptation to metabolic stressors like nonthyroidal illness.
Pilo’s aims and methods

First, here’s the basics of Pilo’s study.
It had nothing to do with deciding what dosage ratios of T4 and T3 hormone were appropriate for thyroid-disabled or thyroidless people. But that’s how it is often misused today, ever since Escobar-Morreale and team proposed clinical trials of thyroid hormone combination therapy based on Pilo’s averages.
Pilo’s team of researchers were trying to understand how much T4 and T3 were secreted and how much T4 became T3 outside the thyroid gland in healthy people with healthy thyroids who had a TSH between 1 and 2 mU/L.
To learn this, the researchers performed an intensive biochemical experiment involving radioactive-iodine-tagged T4 and T3 molecules ([125-I]T4 and [131-I]T3) injected into healthy patients. They attempted to distinguish the conversion of tagged hormones from the non-tagged hormones secreted by the thyroid gland to find out how much came from the thyroid gland.
Combining complicated mathematics, Sephadex gel filtration chromatography, and radioimunoassay testing methods, they measured hormone concentrations.
Based on these measurements, they used theoretical models to estimate each human subject’s global daily T4-T3 conversion rate (CR) and thyroidal secretion rate (SR) of T4 and T3 as fractions of the total amounts of circulating T3 and T4 in bloodstream.
Key flaws in the study

Contemporary thyroid science’s overreliance on this flawed and outdated study, which has never been replicated, is frankly puzzling.
A lot of these scientist’s work was based on refining older theoretical models of thyroid hormone exchange across “compartments” in the human body. Their models were rough. They embedded at least five major inaccuracies and blind spots:
1. Their study pre-dated our modern understanding of transmembrane thyroid hormone transporters and the three thyroid hormone deiodinases. These are two major pillars of the thyroid hormone economy. As a result, every time we cite 20/80, we are returning to this incomplete, outdated metabolic model.
2. There is no sign of any investigation into the subjects’ conversion of T4 into “Reverse T3” (RT3), which would have made their calculations more precise. By verifying the concentrations of the 3rd most abundant thyroid hormone, the most significant “byproduct” of T4 metabolism, their theory-based mathematical estimates of peripheral T3 would have been confirmed.
Measuring both RT3 and T3 is like measuring how much heat and light is produced from a light bulb when measuring its energy efficiency.
Without this simple check and balance, one wonders whether their mass chromatography techniques could have distinguished T3 from RT3 molecules, given that these two hormones have the same molar mass of 650.97 (according to PubChem).
3. They also did not fully take into account the intrathyroidal conversion of T4 into T3 as blood flows through the thyroid (See Berberich et al, 2018), strangely assuming that thyroid “secretion” was synonymous only with “synthesis,” and that “conversion” was only peripheral outside the thyroid gland.
4. They didn’t account for the way TSH hormone can powerfully shift ratios of T3 and T4 synthesis (Citterio et al) and T4-T3 conversion. They didn’t study humans at various TSH levels to observe this conversion-boosting factor in action. They only studied people with TSH in the narrow range from 1-2 pmol/L. They also used a TSH assay that only gave one digit after the decimal point: likely an old, 2nd generation assay.
5. Their subjects were iodine-overdosed every day during the study. This was a common practice in such kinetic studies of the era because it flooded the body with non-radioactive iodine and lowered the percent of injected radioactive iodine that could be recirculated through the thyroid gland as T4 and T3 during the 8-day experiment.
However, acute iodine overdosing also biases T4 and T3 secretion rate from the thyroid–the rate their experiment was trying to discover.
Despite these flaws and others revealed below, the details and insights found within Pilo’s study are still valuable.
One should not throw away gems of truth just because they are rough-hewn. Older studies in thyroid science still have a lot to say.
Findings
Older studies like Pilo’s often provided detailed data tables in their articles, with patient-by-patient data, not just averages and ranges. This detail enables intelligent analysis of human variation. This is invaluable to clinicians who treat individuals, not averages.
Pilo’s article contains many complex tables, like Table 2 shown below. (No, dear reader, you don’t have to read the fine print! I’m just giving you an idea of how much data is in Table 3 alone.)
Data tables like this one form the basis of my reformatted tables below.

From this table, I’ve obtained the Secretion Rates of T4 and T3 (SR-T4, SR-T3) on the right two columns, and the daily Production Rate of T3 (PR-T3) per day, per square meter of body surface. Other data used below come from their additional tables.
Thyroidal T4:T3 Secretion Rates and Ratios

One of the major misrepresentations of Pilo’s article comes from relying on the average thyroidal secretion rates and ratios.
The mere averages do not begin to represent the wide range of diverse data.
In an earlier post, I provided a scatterplot to show how ridiculous it is to imagine that “the” thyroid gland secretes approximately 14 micrograms of T4 for every microgram of T3, an average ratio derived from this study.
Each person’s T4-T3 secretion ratio is a blue dot.
Where is the pattern?
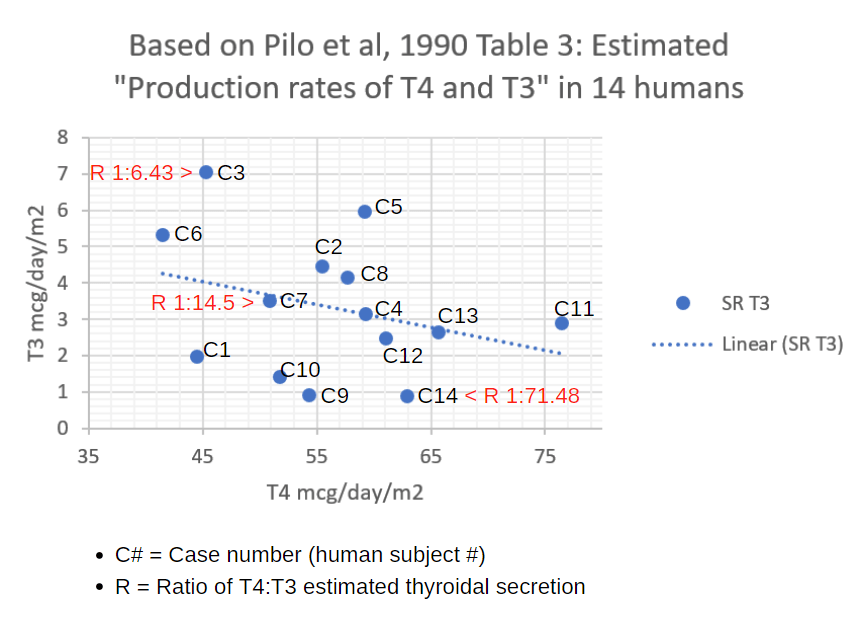
Do you see a central tendency or cluster of dots?
Does the dotted trendline help you see a trend in this constellation?
Case seven (C7) represents a ratio closest to the statistical average ratio of 1 mcg T3 to 14.5 mcg T4. Could you have guessed that dot was the average?
This is a situation in statistics when an average is not meaningful.
Table view
The dots in the scatterplot graph above correlate with the data in the right-hand column in the table below.
As you can see, patient 3 (in row 3 of the table body) has a thyroidal secretion ratio of 6.4 micrograms of T4 per microgram of T3.

Sorting a heat-mapped table by a single column helps one see patterns, if they exist.
This table’s data is sorted by the middle column in green that has the smooth gradation of white at the top to dark green at the bottom: the T4 secretion rate.
The yellow-colored cells in the averages are derived from Pilo’s Table 3 above. The blue averages are calculated from his data using simple math.
As you can see in the graph below, once I multiplied Pilo’s data on the daily thyroidal secretion rate of T4 by the surface area of the 14 patients’ bodies in square meters (given elsewhere by Pilo), I obtained the second green column of data showing these patients’ T4 secretion amount per day.
The average T4 secretion in this small population is not 85 mcg as the ATA said (Jonklaas et al 2014, quoted above), but 99.4 mcg/day.
However, before we jump to the conclusion that we should dose LT4 100 mcg per day to imitate the average daily amount of thyroid secretion, here’s an important fact to remember:
Thyroidal secretion of T4 or T3 is not physiologically or pharmacologically equivalent to oral dosing of thyroid hormone medication.
Let’s consider the adjustments that make today’s standard levothyroxine monotherapy incapable of approximating natural thyroidal secretion.
First of all, the dosing rate per day must be higher than the healthy human secretion rate per day partly because LT4 is poorly absorbed through the small intestine in the GI tract (Wiersinga et al, 2012).
Secondly, because thyroid patients with damaged or missing thyroid glands talking levothyroxine alone have no replacement for their T3 secretion, they need to achieve FT4 levels that are significantly higher than the population average (Jeon et al, 2019). This high-normal FT4 is necessary to maintain enough converted T3 in circulation to produce an euthyroid state when judged by TSH alone.
Even a mildly elevated FT4, once honestly called “chemical hyperthyroidism,” has long been considered acceptable in levothyroxine thyroid therapy (Rendell & Salmon, 1985). It’s essential to provide sufficient thyroid hormone even when FT4 in the top 1/5th of statistical reference range is a risky state associated with “sudden cardiac death” in males (van Noord et al, 2008).
As you can see, our traditions of levothyroxine therapy already prove that endocrinologists treating hypothyroidism cannot make dosing (and its biochemical results) perfectly imitate the average secretion rates and ratios found in untreated human beings.
Endocrinologists have accepted both the abnormality and the health risks within their preferred mode of LT4 monotherapy.
A ratio of 0% T3 from thyroidal secretion to 100% T3 from peripheral metabolism is physiologically incorrect, but it has been deemed acceptable in LT4 monotherapy.
Therefore, limiting either levothyroxine therapy or combination T3-T4 thyroid therapies strictly to the narrow statistical averages of thyroidal secretion found in Pilo’s healthy patients is unacceptably restrictive.
Yet this is exactly what our contemporary guidelines for T3-T4 combination therapy attempt to do (Wiersinga et al, 2012). It is a double standard to permit the unnatural, risky aspects of levothyroxine monotherapy while strictly forbidding all LT3 doses higher than the “physiological” average represented by patient #7 alone.
Bar graph of T3 secretion
Now instead of focusing on T3:T4 ratios and T4 secretion, let’s look at T3 secretion data using a bar graph.
The myth is that 20% of our daily T3 supply is secreted from the thyroid gland.
There is no central tendency in the data.
Which patient comes closest to the 20% average? It’s patient 8 this time.

Therefore, saying “the thyroid secretes 20% of one’s daily T3 supply” is extremely misleading.
Let’s do the endocrine math.
If your thyroid secretes a certain amount of T4 into your blood over a 24 hour period, you may convert anywhere from 16.9% to 42.9% of this T4 into T3 hormone (conversion “rate,” shown later, in graphs below).
This conversion “rate” of micrograms per day per meter of body surface area, when multiplied by each research subject’s body surface area, yields the net “amount” of thyroidal T3 in micrograms per day for each person.
You can then express the “thyroidal” micrograms of T3 as a percentage of their total micrograms of T3 produced per day. That’s what you see in the blue bar graph above.
The thyroid glands in this study contributed between 6.5% to 42.0% of the total daily T3 supply.
That is, you might be lucky to get that much T3 from your thyroid IF your TSH is between 1-2 and you have a healthy thyroid gland, as these 14 subjects did.
Even in a person whose T4 secretion is well regulated by a statistically-average TSH like these people’s was, it’s a roll of the dice how much T3 they’ll convert from their T4 in peripheral tissues. This wide-ranging, individualized conversion rate determines how much T3 their gland secretes daily to compensate.
Continue to Page 2:
- The shifting percent of T3 from peripheral conversion
- The total amount of T3 from peripheral conversion
- How does TSH affect secretion and conversion rate?
- How do sex and age influence these rates?
- What about health factors?
- Solutions?
- References

Leave a public reply here, on our website.