
No, this is not just an article for thyroid science buffs.
Anyone who claims to understand thyroid hormone metabolism should become comfortable using the word “deiodinase.” It’s just as important a word for thyroid disease as “insulin” is for diabetes.
Three enzymes convert thyroid hormones throughout our bodies:
- Deiodinase type 1,
- Deiodinase type 2, and
- Deiodinase type 3.
These enzymes are commonly abbreviated D1, D2 and D3.
The genes that regulate their synthesis are called DIO1, DIO2 and DIO3.
In the past decade, too much emphasis has been placed on genetic polymorphisms — hard-wired mutations — in DIO1 and DIO2 genes, such as the infamous DIO2 rs225014 Thr92Ala polymorphism.
Scientists caution us about the limitations of this perspective on genetic handicaps in thyroid hormone metabolism. For example, deiodinase polymorphisms are limited in the harm they can do to people with healthy thyroids. They become more of a handicap after the thyroid gland is disabled or removed and can no longer compensate for poor T4-T3 conversion during hormone therapy. (See “Thyroid T3 secretion compensates for T4-T3 conversion“)
Studies have just begun to look at the way epigenetic factors can more powerfully alter these enzymes’ function during our lifetime, during illness, and in response to T3-T4 ratio imbalances induced by thyroid hormone therapy.
Here’s a good illustration of the contrast between deiodinase genetics (polymorphisms) and epigenetics (RNA expression):
Recently, in 2016, a team of scientists discovered that the genetic programming for the deiodinases was NOT responsible for depression. They were disappointed to admit their hypothesis was not proven, but they then decided to use different methods to understand the phenomenon better through the lens of epigenetics. In 2017, the same research team published a study that announced that
“peripheral DIO1 and DIO3 expression on mRNA/protein levels may be associated with depressive disorders.”
(Gałecka et al)
We can’t do much about genetics. We CAN do something about epigenetics.
Therefore, understanding how these enzymes’ RNA expression shifts during life, not just how genetic programming is determined at birth, is key.
This helps us to understand why some very puzzling thyroid symptoms (such as depression) respond in puzzling ways to T3 dosing even in people with healthy thyroids.
It also helps us understand many of the normal, expected symptoms of overtreatment, undertreatment and poor conversion in thyroid therapy for hypothyroid individuals.
In this post, you’ll find an explanation of the epigenetic influences on DIO1, DIO2, and DIO3, graphs of tissue RNA expression of these three genes, and discussions of their implications for human health, especially in women.
Genetics vs. epigenetics
Many people are aware that scientists have discovered genetic polymorphisms in DIO1 and DIO2 that make T4-T3 conversion less efficient.
However, having a genetic polymorphism or not is only a small fraction of the significance of these genes.
A large part of the function of these enzymes is regulated epigenetically, by environmental stimuli such as age, lifestyle choices, and disease status.
As explained by the website What Is Epigenetics,
“Epigenetics is the study of heritable changes in gene expression (active versus inactive genes) that do not involve changes to the underlying DNA sequence — a change in phenotype without a change in genotype — which in turn affects how cells read the genes.”
A look at the Human Protein Atlas online presents graphs and data from scientific studies of tissue expression of these enzymes.
You will see that the vast majority of the D1 and D2 enzymes that convert T4 to T3 are likely to be expressed in the thyroid gland itself. This is significant to people who have lost much or all of their functional thyroid tissue.
Another interesting insight is that women and men have a different expression of D3, the enzyme that converts T4 into Reverse T3 and converts T3 into inactive forms of T2.
Which deiodinases are responsible for T4-T3 conversion?
This background section explains how each of the three deiodinases are epigeneitcally upregulated or downregulated.
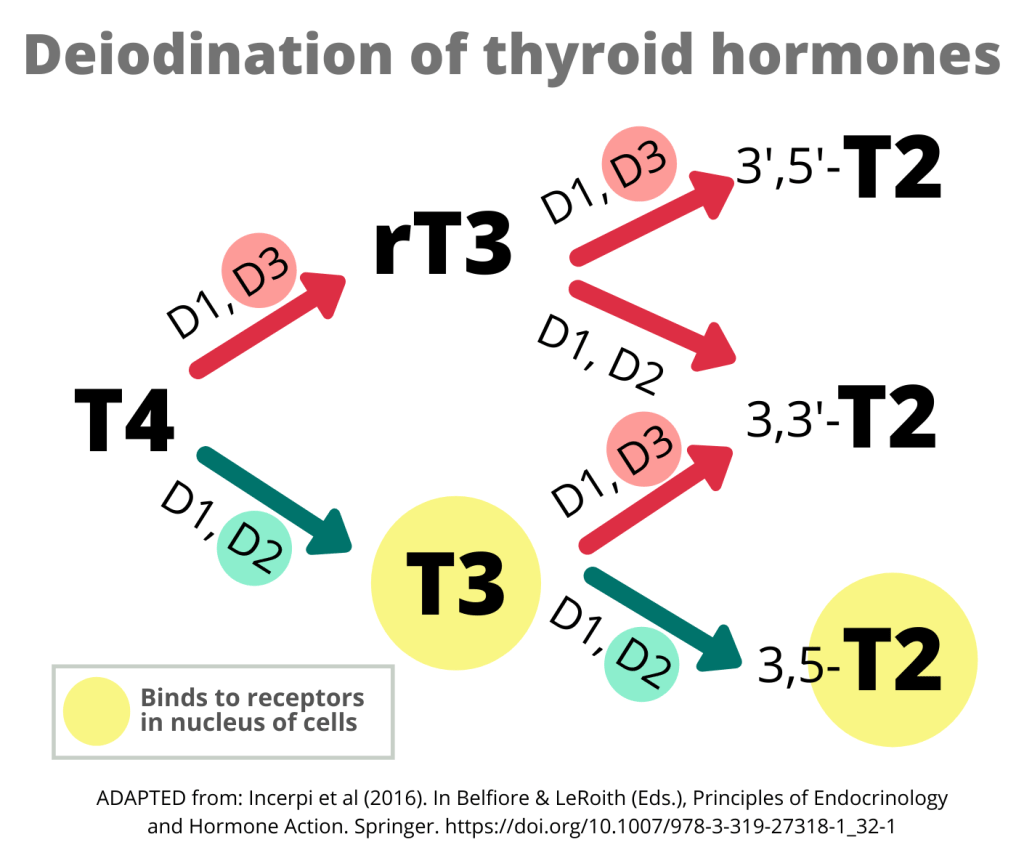
Where do D1, D2 and D3 convert thyroid hormones?
Thyroid hormone conversion happens inside cells, after dedicated transport proteins carry thyroid hormones past the cell membranes into the cells.
D2 is dedicated to T4-T3 conversion
D2 activates the prohormone T4 into the active hormone T3. It also converts T3 into an active form of T2 that can bind to hormone receptors in the nucleus.
What makes DIO2 epigenetically upregulate? Lower levels of T4 in bloodstream and higher levels of TSH, such as the situation found in iodine deficiency and also in early autoimmune thyroid failure.
What makes DIO2 less active? Rising levels of T4 even within reference. The T4 hormone is D2’s main “substrate,” which, in excess supply, overwhelms the enzyme and causes the “ubiquitination” of D2.
The DIO2 gene’s epigenetic response is essential for human survival because it enables us to live on a relatively larger supply of T3 whenever T4 drops lower.
D1 is a two-faced, multi-purpose enzyme.
It is helpful for metabolic flexibility because it can perform all types of thyroid hormone conversion, both “inactivation” and “activation.” DIO1 is located in or near the cell’s plasma membrane and converts hormones as they enter the D1- expressing cell.
What makes DIO1 epigenetically biased toward the T4-T3 conversion path more than T4-RT3 conversion path? Higher T3 levels in blood. This is because DIO1 is unique in containing two thyroid response elements that are sensitive to T3 hormone. (Maia et al, 2011)
What downregulates DIO1? Excess RT3 (this is D1’s main substrate) and the anti-thyroid medication PTU that is used to treat Graves’ hyperthyroidism.
Benefit of epigenetic regulation:
D1 enables us to achieve levels of T3 in the upper half of reference range despite D3 overexpression or D2 repression, conditions desecribed next.
D3 is dedicated to T4-RT3 and T3-T2 inactivation pathways.
D3 is the “firefighter” or soldier team that protects the body against temporary mild excess in thyroid hormone.
Sometimes even “normal” levels can be excessive at the moment, as in the acute phase of a “nonthyroidal illness syndrome” (NTIS), when the body quickly dumps T3 supply to lower the metabolic rate within hours after an acute injury, cardiac surgery, or a heart attack or stroke.
What makes DIO3 epigenetically upregulate its presence in tissues? 1) excess T4 and/or T3 levels above one’s current setpoint, 2) hypoxia caused by obstructed blood flow or pulmonary function, and 3) inflammatory cytokines.
What makes DIO3 calm down and repressed? Lower T4 levels (especially hypothyroidism) and an improving state of health after a crisis. These are both situations in which the body needs to increase its T3 supply to survive a T4 deficiency, or needs to recover from nonthyroidal illness that has depleted T3.
Benefit of epigenetic regulation:
D3 is the first responder to thyroid hormone excess. It can successfully prevent tissue thyrotoxicosis in many subclinical and some overtly hyperthyroid states.
This is the protective element that causes many doctors to be puzzled by the lack of hyperthyroid symptoms in the early stages of severe thyroxine (LT4) overdose.
In fact, the upregulation of this enzyme by T3 itself is likely the factor responsible for “attenuating” the wide fluctuations in T3 levels seen during euthyroid LT3 hormone dosing, as observed by Robert Utiger and colleagues in the 1970s.
All of the deiodinases are based on selenium: they are “selenoproteins.” When you prevent selenium deficiency, you enable all three enzymes to function, including D3, which inactivates thyroid hormones.
However, increasing dosing of selenium past sufficiency does not preferentially aid T4-T3 conversion, because these enzymes are not regulated by selenium but by epigenetic factors such as T3, T4 and TSH levels, and other substances in the body.
Be careful: selenium has a toxicity level and can be overdosed if you take much more than the upper limit of 400 mcg/day. High levels of selenium may be found in some varieties of brazil nuts.
DIO1 and DIO2 RNA expression
In the image are partial screenshots of bar graphs from the Human Protein Atlas. For example, on its DIO1 tissue expression page, the “consensus dataset” graph can be sorted from most to least by clicking on “expression.”

Human Protein Atlas data characteristics
Keep in mind these limitations of the Human Protein Atlas data set:
- The “consensus” data set is in “nX” units denoting the “normalized expression” of levels derived from 3 data banks of tissue samples.
- Human Protein Atlas quantities are per unit of the sample taken from the tissue or organ, not the total amount per organ or tissue. The tissue’s total volume must be considered when assessing its contribution to the body as a whole.
- Tissues were not taken from healthy controls. In many cases, they were from surgeries. Some tissues were adjacent to pathological tissue (i.e. tumors). A person whose tissues were removed during systemic illness, hypothyroidism, or hyperthyroidism may have had abnormal deiodinase expression in many tissues. Nevertheless, samples were examined by experts to determine they had cellular characteristics of healthy tissue.
The prominence of thyroid tissue
As you can see, thyroid gland tissue is the richest in expression of deiodinases type 1 and 2. Although it is an organ smaller in size than many others, its main physiological roles are to create and convert thyroid hormones.
This organ is highly flexible in its supply of hormones to the body partly because of its expression of deiodinases.
TSH stimulation regulates the thyroid’s deiodinases and shifts its T4-T3 conversion rate even when TSH is within the normal reference range (Jang et al, 2020).
It means those who have little to no thyroid tissue will usually convert T4 to T3 less effectively simply because of their thyroid gland disability.
The population of people with impaired T4-T3 conversion includes more than just people who have had a total thyroidectomy. Some people with autoimmune thyroid disease have had complete thyroid fibrosis or atrophy and are equally handicapped as those who have no thyroid at all.
This is something that certain perceptive thyroid scientists have been trying to point out for a long time, as they have demonstrated the existence of the “TSH-T3 shunt.” (Berberich et al, 2018)
In the TSH-T3 shunt, blood carrying TSH enhances T4-T3 conversion as it flows through the thyroid. This is because the thyroid gland is more than just a factory that synthesizes hormones:
The thyroid gland is also a metabolizing organ that converts T4 to T3.
“Within the thyroid, TSH enhances both T3 formation and enzymatic conversion of T4 to T3.”
– Hoermann et al, 2018 “The role of functional thyroid capacity in pituitary feedback regulation”
There is no thyroid hormone pill that can perform this metabolic function of “enzymatic conversion,” and therefore, therapies are limited:
- Thyroid hormone replacement is not a full thyroid gland replacement.
- Thyroid hormone replacement is not a DIO1 or DIO2 enzyme replacement.
- A daily T4 hormone pill is utterly incapable of ensuring its sufficient activation to T3 in the body.
A thyroidectomy is not a risk-free solution for a “good thyroid cancer” if it steals these enzymes from one’s body, unreasonably demanding the liver and kidney to suddenly convert more T4 than they ever did before.
Our bodies are less effective at activating T4 to T3 when there is less thyroid gland tissue, when T4 is higher in reference, when T3 is lower in reference (this means a low Free T3:T4 ratio), and when TSH is lower.
Blood thyroid hormone levels: D1 or D2?
Prior to approximately 2005, the widespread belief was that D1 activity was responsible for most of our circulating T3 because liver and kidney were observed to be a “compartment” that quickly exchanged thyroid hormone, and they both expressed a lot of D1.
However, the scientific community became skeptical of this DIO1 dependency because
- D1 is predominant in only a few tissues, while D2 is predominant in many, and
- At “normal” concentrations of T3 and T4 and TSH, the D1 enzyme converts T4 into Reverse T3 about 50% of the time. (Maia et al, 2011)
However, now the pendulum should be swinging back again.
It now seems that from some reports, even DIO1 genetic hard-wiring is actually more responsible for blood levels of T3 after all.
- In 2008, Panicker et al did a research study showing that when a person has a DIO1 polymorphism, either rs2235544 C>A or rs11206244 C>T, it can reduce enzyme activity to such a degree that the Free T3:T4 ratio is lower.
- In contrast, later studies have discovered that when a person has a DIO2 polymorphism, rs225014 T>C, it is associated with health problems, but bloodstream Free T3:T4 thyroid hormone ratios are not significantly affected. (Panicker et al, 2009).
Therefore, DIO1 seems to be more responsible for circulating T3 after all, even at a genetic hard-wiring level, even outside of thyroid disease.
The importance of protecting serum levels of T3
Certain thyroid therapies, as well as nonthyroidal illnesses, can short-circuit our body’s ability to epigenetically enhance DIO1 and DIO2. They fail to protect T3 levels in blood.
Bianco et al, in a recent comprehensive review (2019), have emphasized the importance of maintaining healthy levels of T3 in circulation, and not overrelying on local T4-T3 conversion:
“in most tissues, the level of TR [thyroid receptor] occupancy, expression of T3-responsive genes, and downstream biologic effects are greatly influenced by circulating T3 levels.
In other words, as long as TH [thyroid hormone] transmembrane transporters are available, T3 from plasma will enter cells at levels that occupy half of the TR pool.
Conversely, a drop in plasma T3 will reduce TR occupancy in most tissues as well.”
– Bianco et al, 2019
Consider this fact, dear American Thyroid Association guideline-writers, the next time you are tempted to make excuses for LT4’s reduction of patients’ plasma T3 supply.
Having TSH in the normal range is not a fair trade for T3 loss.
T4 monotherapy uses all three levers to epigenetically reduce both DIO1 and DIO2 RNA expression:
- 1) elevating FT4 levels significantly above those found in the normal healthy population limits DIO2 expression,
- 2) depressing FT3 levels significantly below those in the normal healthy population limits DIO1 expression, and
- 3) causing TSH to fall abnormally lower per FT3 unit, due to artificially raised FT4, limits DIO2 expression.
How dare anyone read Bianco’s quote above and then continue to dismiss a treated thyroid patient’s chronically low or “mildly subnormal” T3 levels as benign, or say they don’t know of any adverse effects and that therefore there must be none?
Dear gullible endocrinologists who promote UNwise Choosing Wisely campaigns to prevent so-called “unnecessary” FT3 testing in treated thyroid patients:
- How can you believe that D1 and D2 are still functioning normally in cells throughout their body despite what science says, and despite the obvious suppression of Free T3:T4 ratios and FT3 levels seen in publications like Gullo et al, 2011?
- Have you asked a low-T3 thyroid patient how they are feeling while living on the edge of tissue hypothyroidism for the sake of a mandated pituitary secretion of TSH that can’t stimulate their dead or missing thyroid?
- Have you discovered how many of your T3-poor patients are submerged below the level of T3 supply in brain or heart?
No? Maybe it’s because your campaign is designed to forbid the cheap $10 test to discover our low Free T3 levels so that someone can rescue us. Your campaign is so evidence-blind that you forbid doctors from identifying who needs rescuing. If doctors can’t understand how D1 and D2 are both epigenetically DOWNregulated, and if they can’t divide FT3 by FT4 and examine thyroid hormone levels in the context of symptoms, they simply don’t know how to distinguish an underdose from a drug-induced state of poor T4 conversion. Doctors have no idea who on TSH-normalized LT4 monotherapy is being subjected to a pharmaceutical T3-ectomy.
DIO3 RNA expression

This RNA map is the most surprising and revolutionary of all.
For many decades, it was stated that “This enzyme is present in brain, skin, placenta, and fetal tissues of the rat” (Escobar-Morreale et al, 1997) and therefore the belief was that most adult human tissues did not express D3 either.
Doctors, revise such statements.
Notice the updated consensus measurements per tissue type as of 2020.
Where are the DIO3 expressing tissues in adults during a state of health? Not just in brain and skin.
As you can see, RNA expression of D3 is extremely prominent in
- female reproductive tissues, most of all the cervix and uterus, followed by
- the placenta,
- the retina of the eye,
- lymph nodes, and
- urinary bladder.
Next comes D3 RNA expression in
- body fat (adipose tissue),
- the insulin-secreting organ of the pancreas,
- the thyroid gland,
- and three more female tissues.
How much scientific study has been investigating the high level of D3 expression in female reproductive tissues beyond the condition of pregnancy? Very little.
- How does this DIO3 expression affect diseases of the retina in the eye?
- How might DIO3 be involved in urinary incontinence in aging?
- How is DIO3 involved in lymphedema or in other disorders of lymph nodes?
- How about DIO3 in female sexual dysfunction or … breast cancer?
- How does DIO3 in fat tissue work against weight loss in obese hypothyroid patients?
- How does this tissue expression of DIO3 RNA affect women with PCOS or endometriosis?
The vast majority of people afflicted with hypothyroidism are females beyond reproductive age.
Researchers, don’t be sexist, ageist, or discriminate against people with a thyroid disability. Study people who are vulnerable to excess thyroid hormone inactivation.
Thyroid patients say:
- STUDY DIO3 RNA expression’s influence on thyroid levels in females with nonthyroidal illnesses of reproductive tissues!
- And vulnerable females on thyroid hormone therapy, please, not just the thyroid-healthy.
- And not just in pregnant women! Also in puberty and post-menopausal women.
References
Click to reveal reference list
Berberich, J., Dietrich, J. W., Hoermann, R., & Müller, M. A. (2018). Mathematical Modeling of the Pituitary–Thyroid Feedback Loop: Role of a TSH-T3-Shunt and Sensitivity Analysis. Frontiers in Endocrinology, 9. https://doi.org/10.3389/fendo.2018.00091
Bianco, Antonio C., Alexandra Dumitrescu, Balázs Gereben, Miriam O. Ribeiro, Tatiana L. Fonseca, Gustavo W. Fernandes, and Barbara M. L. C. Bocco. 2019. “Paradigms of Dynamic Control of Thyroid Hormone Signaling.” Endocrine Reviews 40 (4): 1000–1047. https://doi.org/10.1210/er.2018-00275.
Escobar-Morreale, Héctor F., M. Jesús Obregón, Arturo Hernández, Francisco Escobar del Rey, and Gabriella Morreale de Escobar. 1997. “Regulation of Iodothyronine Deiodinase Activity as Studied in Thyroidectomized Rats Infused with Thyroxine or Triiodothyronine.” Endocrinology 138 (6): 2559–68. https://doi.org/10.1210/endo.138.6.5212.
Gałecka, Elżbieta, Anna Kumor-Kisielewska, Agata Orzechowska, Michael Maes, Paweł Górski, and Janusz Szemraj. 2017. “Assessment of Type 1 and Type 3 Deiodinase Expression Levels in Depressive Disorders.” Acta Neurobiologiae Experimentalis 77 (3): 225–35.
Gałecka, Elżbieta, Monika Talarowska, Michael Maes, Kuan-Pin Su, Paweł Górski, and Janusz Szemraj. 2016. “Polymorphisms of Iodothyronine Deiodinases (DIO1, DIO3) Genes Are Not Associated with Recurrent Depressive Disorder.” Pharmacological Reports: PR 68 (5): 913–17. https://doi.org/10.1016/j.pharep.2016.04.019.
Gullo, Damiano, Adele Latina, Francesco Frasca, Rosario Le Moli, Gabriella Pellegriti, and Riccardo Vigneri. 2011. “Levothyroxine Monotherapy Cannot Guarantee Euthyroidism in All Athyreotic Patients.” PLoS ONE 6 (8). https://doi.org/10.1371/journal.pone.0022552.
Hoermann, Rudolf, John E. M. Midgley, Rolf Larisch, and Johannes W. Dietrich. 2018. “The Role of Functional Thyroid Capacity in Pituitary Thyroid Feedback Regulation.” European Journal of Clinical Investigation 48 (10): e13003. https://doi.org/10.1111/eci.13003.
Jang, D., Marcus-Samuels, B., Morgan, S. J., Klubo-Gwiezdzinska, J., Neumann, S., & Gershengorn, M. C. (2020). Thyrotropin regulation of differentiated gene transcription in adult human thyrocytes in primary culture. Molecular and Cellular Endocrinology, 518, 111032. https://doi.org/10.1016/j.mce.2020.111032
Maia, Ana Luiza, Iuri Martin Goemann, Erika L. Souza Meyer, and Simone Magagnin Wajner. 2011. “Type 1 Iodothyronine Deiodinase in Human Physiology and Disease.” Journal of Endocrinology 209 (3): 283. https://doi.org/10.1530/JOE-10-0481.
Panicker, Vijay, Christie Cluett, Beverley Shields, and Anna Murray. 2008. “A Common Variation in Deiodinase 1 Gene DIO1 Is Associated with the Relative Levels of Free Thyroxine and Triiodothyronine.” The Journal of Clinical Endocrinology and Metabolism 93 (8): 3075–81. https://doi.org/10.1210/jc.2008-0397.
Panicker, Vijay, Ponnusamy Saravanan, Bijay Vaidya, Jonathan Evans, Andrew T. Hattersley, Timothy M. Frayling, and Colin M. Dayan. 2009. “Common Variation in the DIO2 Gene Predicts Baseline Psychological Well-Being and Response to Combination Thyroxine plus Triiodothyronine Therapy in Hypothyroid Patients.” The Journal of Clinical Endocrinology and Metabolism 94 (5): 1623–29. https://doi.org/10.1210/jc.2008-1301.

Leave a public reply here, on our website.