Page 2
Back to Page 1
Flip-flops between hyper and hypo
It is rare for thyroid status to flip-flop to its opposite, but it does happen.
In Takasu and Matsushita‘s 2012 study,
- 2 (5.8%) of the 34 HYPOthyroid people became HYPERthyroid.
- 2 (2.0%) of the 98 HYPERthyroid people became HYPOthyroid.
Alzahrani et al in 2005 published a case of thyroid status fluctuation that revealed a three-phase fluctuation from hypo to hyper, then again to hypo status, as shown in their graph:
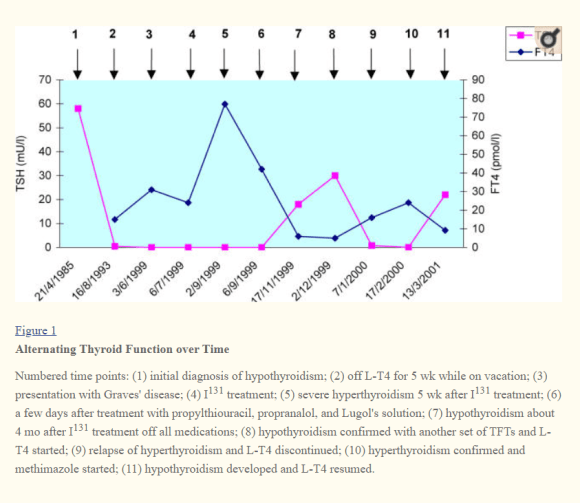
When the patient represented by the graph was hypothyroid, TPOAb were found to be elevated, but unfortunately TBII was not tested. The authors could only theorize that she could have had active TBAb causing hypothyroidism, and they cite Takasu’s works to support this theory.
While this case and many others show large fluctuations over a short term, long-term stability can be deceptive. Another case of hypo to hyper occurred 27 years after the hypothyroid diagnosis and stable thyroid therapy (Ahmad et al, 2018). My biochemical shift to more extreme hypothyroidism occurred after about 13 years of therapy.
Lesho & Jones’ 5 case studies
In 1997, Lesho and Jones published an article with five case studies of patients going through what I call “hypo-hyper flip-flops,” describing them in enough clinical detail to be enlightening to a patient and physician:
Case 1, a 26-year-old woman, changed from hypo to hyper to hypo (3 states). She began with a small goiter, TPOAb, and a TSH of 30 mU/L. After five years on levothyroxine, her TSH suddenly became suppressed, and both TSI and TBII tests were positive. After LT4 medication was stopped, TT4 fell from 12 to 4.3 mcg/dL. Three years later, TSH had risen to 82.4 mU/L and she had positive TBII results, this time, likely due to TBAb. She was given LT4 therapy again. She moved away and was lost to follow-up.
Case 2, a 28-year-old woman, cycled from hypo to hyper to hypo to hyper (4 states). She first presented with a TSH over 100 mU/L, high TPOAb, and “no palpable thyroid tissue” on examination, and was placed on LT4. Four months later, she had a small goiter, signs and symptoms of hyperthyroidism, a suppressed TSH, mildly elevated TT4 of 12 mcg/dL, and a positive TSI test result. She had a positive pregnancy test result at the time, so she was simply taken off LT4 and monitored rather than being given anti-thyroid medications. She then had a “brief episode” of hypothyroidism with TSH of 78 mU/L. Following that phase came “florid hyperthyroidism and a diffusely enlarged thyroid (four times normal size).” She was then treated with PTU, and later with a combination of PTU and LT4. Finally, she underwent radioiodine ablation of her thyroid gland.
Case 3, a 22-year old woman, cycled from hyper to hypo to hyper (3 states). She had a family history of thyroid disease. Shehad been on lithium therapy for bipolar disorder 6 months before her records showed hyperthyroidism with a TSH of <0.2 and TT4 of 14 mcg/dL. She had symptoms consistent with biochemistry, but without therapy. Six months later, she presented to the authors of this article with a TSH of 73 mU/L and a TT4 of 2.5 mcg/dL, a “firm, enlarged thyroid (twice normal size).” After 3 months of LT4 therapy, she had become “overtly hyperthyroid” with suppressed TSH and a TT4 of 14 mcg/dL. “On examination, the thyroid had further enlarged to three times normal size, with a soft bruit audible over both lobes.” LT4 was withdrawn. She was later treated with radioiodine ablation.
Case 4, a 25-year-old woman, went from hypo to hyper to euthyroid, (3 states) and then continued to fluctuate at least 3 more times. Upon diagnosis she had TSH >100 mU/L and was put on LT4 therapy. Six months later, she reported having “progressive weight loss, insomnia and tremor.” The authors report that “Routine thyroid tests showed hyperthyroidism, and radioiodine uptake was 50%.” After LT4 was removed, “the patient quickly became euthyroid.” Next, “during close follow-up over the next 5 years, she had three distinct spontaneous fluctuations from hyperthyroidism to hypothyroidism. Tests for TSI during her hyperthyroid periods were clearly positive.” She then declined further therapy and was lost to follow-up.
Case 5, an 18-year-old man, presented as hyper, then hypo, then hyper (3 states). At first, he had “a goiter and elevated thyroid hormone levels. Radioiodine uptake at 24 hours was 63%.” He was treated with PTU and propranolol. Only two months later, he returned, complaining of fatigue. The TSH was slightly elevated at 8.1 mU/L and TT4 was low at 3.2 mcg/dL. PTU was discontinued after tests for TSI were negative but TBII was positive at 47.8% (normal is <10%). Next, three months later, he was again thyrotoxic with a RAI uptake of 59%, and he underwent radioidine ablation.
Misunderstanding persists
Unfortunately, ignorance persists regarding such wide fluctuations, despite many publications.
As of 2018, some doctors were still publishing case studies revealing ignorance that TSAb and TBAb can be responsible for extrathyroidal manifestations in eyes or thymus as well as obvious changes in thyroid status (An et al, 2018).
Clearly, such transformations are frustrating and distressing for both the patient and the doctor, and they can lead to harm to the patient.
If the patient gives up on treatment or moves away, as described at the end of some of these case studies, they may engage another doctor without transferring medical records or knowledge to them.
Both the patient and the doctor ought to be aware of what is going on and why, when these significant transformations occur.
Hashitoxicosis versus transient TSAb
The occasion of transient thyrotoxicosis in Hashimoto’s thyroiditis has been theorized under the term “Hashitoxicosis.”
Hashitoxicosis can easily be confused with transient Graves hyperthyroidism if the proper antibody test is not done.
As clearly explained by Schaffer, Puthenpura and Marshall (2016),
- “Rarely, HT can present with Hashitoxicosis, which is a transient form of thyrotoxicosis that results from release of preformed thyroid hormone due to inflammatory destruction of thyroid cells. As inflammation resolves and because thyroid hormone release is not due to ongoing stimulation of TSHR, resolution typically occurs within a few months. It is usually asymptomatic, with typically only mild clinical symptoms of thyrotoxicosis if present.”
Confirmation of Hashitoxicosis relies on key features described by Schaffer, Puthenpura and Marshall (2016):
- Elevated thyroid hormone levels and low or suppressed TSH
- “The presence of HT antibodies” (TPOAb and TGAb) – signs of thyroid autoimmunity are present.
- “GD antibodies remain negative” (TBII or TSI or TRAb test negative, revealing not even low levels of TSAb are responsible)
- “mild course associated with absence of clinical symptoms and signs”
However, not every physician or researcher is aware that TSAb need to be ruled out for hyperthyroidism to be “Hashitoxicosis.”
Some cases of so-called Hashitoxicosis in the literature and clinical practice appear to be poorly diagnosed or misunderstood cases of transient Graves’ disease TSAb antibody activity.
For example, In 2014, Kirmizibekmez et al published a case study of a 14-year old girl who had Graves’ eye disease (cause by TSAb), elevated thyroid hormones, and a positive TRAb test result of 21, and yet they called her condition “Hashitoxicosis” just because her TSAb had disappeared seven months later.
But Kirmizibekmez’s case study was not about a Hashimoto’s patient with a transient hashitoxicosis episode. Rather, she had a short-term flare of Graves’ disease that was quickly quenched by Methimazole medication. Given that she could produce TSAb, she had the genetic predisposition for Graves disease (TSAb and/or TBAb).
Therefore, her hyperthyroid episode should have been considered part of her Graves’ disease, not merely a thyrotoxic episode caused by thyroid tissue damage in a pure case of Hashimoto’s.
This is not merely a quibble in terminology.
Misunderstanding led to a possibly unnecessary partial thyroidectomy for a case called “hashitoxicosis” (Wagner et al, 1995). Wagner’s brief English abstract is available for their French article that I have not been able to retrieve. It nevertheless reveals a lot of information.
- After the man’s partial thyroidectomy, skin manifestations of Graves’ disease appeared in the patient, who, by the time he arrived in the care of Wagner et al, had undescribed “anti-thyroid antibodies.”
- His skin manifestations and antibodies seem to have been interpreted as a case of overt hyperthyroidism, since his doctors decided to remove the remainder of his thyroid gland.
- At least this finished thyroidectomy revealed to the doctors that the remaining thyroid gland “stump” had clear signs of Atrophic Thyroiditis.
Their article’s abstract reveals their astonishment and subsequent education: “This unusual development towards atrophic thyroiditis raises the problem of the relations between the various forms autoimmune thyroiditis.”
Understand the full spectrum
Clearly, thyroid autoimmunity represents a spectrum of overlapping autoimmune diseases, not a set of two or three mutually exclusive or static conditions.
Each of us can travel along the full spectrum. You don’t know where you’re destined to go.
This research may provide hope for many patients.
It shows that it is possible for remissions to occur in many hyperthyroid patients and some hypothyroid patients
The research should make us vigilant.
Doctors and patients should not think it’s impossible for several cycles of hyper-hypo transformation to occur over months, years, or decades of therapy.
We should become more thoughtful about how language (mis)categorizes us.
We can’t truly say that TPOAb and TGAb are exclusively “Hashimoto’s antibodies” (autoimmune hypothyroidism) because Graves’ patients have them, too. Nor can you truly say that the two TSH-Receptor antibodies (TSAb and TBAb) are only Graves’ Disease antibodies.
This way of seeing thyroid autoimmunity can help patients identify with peers who are different from themselves.
Science should persuade medical systems about the cost-effectiveness and health benefits of more thorough thyroid antibody testing.
Many of us have been left without a definitive diagnosis of “autoimmune thyroid disease” because of our laboratory’s notes telling the doctor to dismiss our TPOAb and TGAb results. The laboratory notes say these antibodies are also found (but they fail to say less commonly found), outside the autoimmune thyroid disease population, as shown in the table.
However, if we test positive for either TRAb antibody, there is absolutely no doubt. These antibodies are not found at all outside of autoimmune thyroid disease. They provide a definitive “autoimmune thyroid” diagnosis, if we are among the 0-20% of Hashimoto’s patients or 73-100% of Graves patients who have it.
With an autoimmune diagnosis comes the motivation to become informed about the other autoimmune diseases that are most commonly associated with thyroid autoimmunity. I encourage you to do your own research, and in time, I’ll publish more on the topic.
Tips: Do you still need thyroid medication?
The incomplete dissemination of TRAb scientific knowledge also means that some hypothyroid patients may be needlessly continuing thyroid therapy medication for decades, when they could indeed wean off it and live without it if their TBAb-induced hypothyroid phase is now behind them.
What can you do if you are being treated for hypothyroidism, and suspect you might be in remission from hypothyroidism?
Thyroid hormone therapy may now be unnecessary, but that does not mean it is harming you.
Before jumping into medication withdrawal, realize that there is no evidence of any harm to the thyroid gland or to human health if a person is maintained on thyroid therapy while they still have a healthy thyroid gland—as long as they are not overdosed and their thyroid hormone levels are optimized and monitored.
- You’re not necessarily overdosed. Thyroid medication usually lowers TSH, and therefore thyroid hormone therapy can reduce and replace thyroidal secretion—it does not cause thyrotoxicosis to add some thyroid hormone on top of a lower level of thyroidal secretion.
- Hormone medication is not making your thyroid lazy. The thyroid gland is not a muscle, but an endocrine gland. Thyroids continue to secrete at a low level even in the absence of TSH.
Exercise caution before withdrawing medication
Nevertheless, it is not wise to experiment willy-nilly with medication withdrawal for hypothyroid patients on therapy—patients have complex, stressful lives to manage and often have concurrent health conditions that require thyroid hormone stability and optimization.
Patients should not be forced to endure the risk of even temporary hypothyroidism simply to satisfy curiosity.
Even if the thyroid is healthy, it can take a while for TSH to resume its appropriate level of thyroid stimulation. While the pituitary takes 6 weeks to 3 months to raise and fine tune its TSH secretion, a patient may suffer an inappropriately low level thyroidal secretion while underdosed.
Therefore, patients who are currently hypothyroid and on thyroid therapy should not simply cease therapy to discover whether they can live without it. You risk of health and quality of life reductions if the thyroid gland has been too damaged with fibrosis or atrophy to support health on its own.
Do a thyroid ultrasound
If the gland seems perfectly normal upon palpation, and if there was no record of any major gland health problems, maybe it is healthy.
Ultrasounds usually report on the “echogenicity” of the thyroid. Good echogenicity is a sign of the health of thyroid tissue, whereas “hypoechogenic” glands have more fibrosis or damage (Höfling et al, 2008).
If the gland is very small or difficult to detect upon palpation, the thyroid gland’s health should be examined by ultrasound because it could reveal the presence of significant gland atrophy, as well as unsuspected cancers, which are more commonly found in low-volume glands (Duran et al, 2014).
Make sure to ask the ultrasound technician for measurements of the thyroid gland’s size and compare them with population graphs such as those found in Carle et al, 2009. This article revealed that extremely small and large glands are found only in autoimmune thyroid disease. One should understand how abnormal a gland of less than 5 mL truly is by considering Carle’s sample.
Test 50% reduction, not complete withdrawal
Takasu and colleagues suggest that if the hypothyroid patient’s thyroid gland seems healthy, one could cut the LT4 dose in half (i.e. from 100 mcg /day to 50 mcg/day), and then observe and continue to reduce if the response is positive.
Follow up as soon as necessary with a lab test. If the patient becomes symptomatically hypothyroid (low heart rate, low body temperature, cognitive difficulty, extreme fatigue), there is no need to extend their suffering by waiting 6 weeks for TSH to “equilibrate.”
Consider a half-withdrawal in light of the extreme: Full withdrawal studies show that a fully suppressed TSH is the most delayed in rising, but it starts rising on day 3 and can climb to high levels within 7 days in people who do not have thyroid glands (Ross & Cohen, 1990).
As TSH secretion rises, it will produce more FT4 from a healthy gland, until FT4 reaches a healthy equilibrium and TSH reduces; this is the natural process of homeostasis over time. The change is less dramatic if TSH is not fully suppressed, but FT4-TSH relationships should reveal either some thyroid function or no thyroid function by the end of 3 weeks.
Locate and order a TBII test
Ideally, if further confirmation is needed before a dose-reduction experiment, a TBII test may still be found in the patient’s region (Diana et al, 2017-2019 have been advocating for their return to clinical practice).
The TBII test will reveal whether either TBAb or TSAb antibodies are currently active. Do not trust a “TRAb test” to reveal blocking antibodies; such assays are designed and calibrated mainly with hyperthyroid cases in mind. When testing TBII, one should be aware that negative results can still be false negatives for a Graves’ disease diagnosis, since TBAb may have been present upon hypothyroid diagnosis but later disappeared.
Consider Graves’ genetics
Another way to confirm other evidence of a potential Graves’ disease diagnosis in a Hashimoto’s patient is via a consumer genetics test such as 23andme.
Order the full data set of 600,000+ SNPs that they can provide. The “raw data” file can be analyzed through a cheap online service like Promethease.com. The patient can look for her own Graves’ Disease risk SNPs, supplemented by searching for SNP data recently published.
Discover how many polymorphisms indicate a significant risk and make Graves’ a likely diagnosis, compared to the number of polymorphisms you lack, or do not have in your 23andme data file.
My own genetic test results that speak Graves’ susceptibility loud and clear are consistent with my ultrasound evidence of past Graves’ antibody activity. My genetic record confirms the clear diagnostic feature of my atrophied thyroid, because high titres of the TBAb antibody are known to cause permanent “hypothyroid Graves” – Atrophic Thyroiditis.
Once a patient is on no therapy, the free SPINA-Thyr app can be used to assess thyroid gland health and TSH secretion appropriateness on the basis of FT4-TSH data.
My best wishes
There is no hope of remission for my 0.5 mL atrophied gland, but I look forward to learning that others have safely discovered remission, or avoided the harmful mistakes in therapy I experienced, with the aid of scientific understanding.
I’ll be following up with one or more posts already in development that examine the etiology of TRAb and effects of TRAb in the human body, such as their effects on TSH secretion, T4-T3 conversion, and thyroid gland health status.
By Tania S. Smith

Leave a public reply here, on our website.